Innov Clin Neurosci. 2026;23(1–3):32–38.
by Humphrey Masimba Makumbirofa, MSc; Tariro Tsitsidzashe Hluyo, BPharm; Jonathan Chaweza, BTech; Simbarashe Wandayi, MVPG, BVSc; and Nyarai Desiree Soko, PhD, MSc
Messrs. Makumbirofa, Hluyo, Chaweza, and Dr. Soko are with Department of Pharmaceutical technology, School of Allied Health Sciences, Harare Institute of Technology, Harare, Zimbabwe. Mr. Wandayi is with Department of Pathobiology, Faculty of Veterinary Science, University of Zimbabwe, Harare, Zimbabwe.
FUNDING: This research was funded by the Harare Institute of Technology.
DISCLOSURES: The authors have no relevant conflicts of interest.
ABSTRACT: Objective: We describe the development of a probiotic-infused carbamazepine dosage form. This dosage form combines carbamazepine with probiotics, proposing the potential use of both active pharmaceutical ingredients in a single dosage form. Design: Evaluation of Bifidobacterium bifidum and Lactobacillus acidophilus susceptibility to carbamazepine using agar disk diffusion revealed the B. bifidum was resistant to carbamazepine, prompting its selection for development of the dosage form. A suspension was formulated using the precipitation method with stability testing assessing organoleptic properties, pH, and viability of the bacteria in the dosage form. Toxicity assessment was done using 20 male albino Wistar rats, measuring weight, urination, and anxiety changes postadministration of carbamazepine and probiotics, following ethical approval from the University of Zimbabwe’s animal house laboratory and board. Results: Rats receiving only B. bifidum exhibited heightened anxiety and reduced weight, while those receiving carbamazepine alone increased weight and urination but lowered anxiety. Combining carbamazepine with B. bifidum significantly decreased anxiety (P=0.0312) and increased urination, with significant weight reduction (P=0.03186). The dosage form maintained stability, preserving B. bifidum viability over a period of 28 days. Conclusion: Integrating probiotics into carbamazepine formulations offers a promising strategy, with potential applications to other antiepileptic drugs. Keywords: Carbamazepine, probiotics, Bifidiobacterium bifidum, dosage form, infused
Introduction
Carbamazepine is indicated for use as an anticonvulsant drug. Evidence supporting its efficacy as an antiepileptic drug (AED) was derived from studies that enrolled patients with partial seizures with complex symptomatology (psychomotor, temporal lobe), generalized tonic–clonic seizures (grand mal), and mixed-seizure patterns, including the latter 2 types or other partial or generalized seizures.1,2 Absence (petit mal) seizures do not appear to be controlled by carbamazepine. The mechanism of action of carbamazepine remains unknown, but its anticonvulsant activity may result from use-dependent blockade of voltage-sensitive sodium channels.1,2 Carbamazepine has demonstrated anticonvulsant properties in animals with electrically and chemically induced seizures, and it reduces or abolishes pain induced by stimulation of the infraorbital nerve in animals.1,2
Recent studies have shown a link between the gut microbiome and neurological diseases, including epilepsy. Although there have been only a few studies to date that have reported alterations in the gut microbiome in human epilepsy,3,4 the associations between epilepsy and infection, inflammation, and antibiotic treatment raise the question of whether alterations in microbial communities under those conditions may be involved in its pathophysiology.5 A study by Wang et al6 provided evidence that the addition of probiotics in the treatment of patients with temporal lobe epilepsy can effectively enhance the antiseizure effects and alleviate cognitive impairment, neuropsychiatric symptoms, and overall quality of life to a certain degree. The functional activities of the brain depend on the signal transmission between different types of neurons and glial cells, which depends on neurotransmitters; for example, an imbalance of excitatory and inhibitory neurotransmitters may lead to epileptic seizures.7 The intestinal microbiota has a profound influence on several neurotransmitters and neuromodulators, such as monoamines, serotonin, gamma-aminobutyric acid (GABA), and brain-derived neurotrophic factors.8 Different bacterial strains may synthesize different neurotransmitters. GABA is secreted by certain strains of Lactobacillus and Bifidobacterium. Conversely, norepinephrine is produced by certain strains of Saccharomyces, Bacillus, and Escherichia.9 Serotonin comes from Enterococcus, Streptococcus, Escherichia, and Candida,10,11 dopamine from Bacillus and Serratia,7 acetylcholine from Bacillus and certain lactic acid bacteria strains,7 and glutamate from various coryneforms and lactic acid bacterial strains.10,11 Some studies have demonstrated that mice or rats fed with probiotics showed altered neurotransmitter composition and changes in their target receptor throughout different brain regions.12 Interestingly, certain bacterial taxa in the gut produce certain enzymes that can facilitate the synthesis of short-chain fatty acids (SCFAs) by fermentation. SCFAs exert their functions on the central nervous system by binding to the G receptors on entero-endocrine cells.13 This induces the secretion of diverse hormones and neurotransmitters, indirectly influencing brain neurochemistry. One example is acetate, which alters the levels of glutamate, glutamine, and GABA in the hypothalamus. These neurotransmitters play a crucial role in the development of neurological disorders.14
Probiotics are generally regarded as safe; however, due to their increasing consumption and their potential to influence metabolism, the efficacy and safety of orally administered probiotics deserve a closer look.15 These organisms act by affecting absorption, composition, or metabolic activity of the gut microbiota, potentially altering the bioavailability of the drug.16 Probiotics can influence bioavailability of drugs by several possible mechanisms, including alterations of gastrointestinal properties like pH, increasing intestinal transit time,17 or thickening adherent mucus and altered expression of intestinal transporters across the intestinal wall. In addition, they work by inducing or inhibiting microbial enzymes, thus modulating their activity on drugs and by using drugs as surrogate substrates for their growth.15 Effective amounts of a probiotic when combined with an effective amount of drug tend to result in enhanced therapeutic outcomes.18 Developing a single product that combines carbamazepine with probiotics as a unified dosage form has the potential to reduce the burden of polypharmacy by improving patient adherence to medication since it is easy to administer. Thus, this study aimed to develop a probiotic-infused carbamazepine dosage form for the potential use in the management of epilepsy.
Materials and Methods
Viability testing of microbial strains in carbamazepine. The susceptibility of the potential probiotic cultures to carbamazepine was determined using the disk diffusion method as per recommendations from the National Committee for Clinical Laboratory Standards.19 Isolates of Lactobacillus acidophilus and Bifidobacterium bifidum were obtained from the Department of Biotechnology at the Harare Institute of Technology. A range of carbamazepine (Datlabs) concentrations were prepared—4 mg/mL, 5 mg/mL, 8 mg/mL, 10 mg/mL, and 12 mg/mL, as per the British Pharmacopoeia guidelines.20 The disk diffusion method was used to determine the susceptibility of each selected species to different concentrations of carbamazepine. B. bifidum was selected for development of the probiotic-infused carbamazepine based on the bacteria’s ability to grow in carbamazepine.21 Standard plate count using Lactobacillus MRS agar (HiMedia Laboratories) was performed as according to Fowler et al22 to estimate the number of bacteria present. The colony forming units (CFU) per gram of the original B. bifidum sample that formed on the agar plates were counted by selecting the plate which had about 25 to 250 colonies.23 This provided an estimation of the bacterial load before further experimental work commenced.
Evaluation of combined effect of probiotic and carbamazepine in animal models. Ethical approval to evaluate the combined effects of probiotics and carbamazepine in animal models was obtained from the University of Zimbabwe Animal House. A total of 20 male albino Wistar rats (body weight :190–210 g; age: 7–8 weeks) were purchased from the University of Zimbabwe Animal House. All animals were fed a standard diet of rat pellets and water ad libitum. After an adaptation period of 7days, the rats were randomly allocated to different treatment groups.
The rats were divided into 4 groups. The control group (n=4) received no therapeutic intervention. The B. bifidum group (n=4) was administered 1 g of B. bifidum probiotics dissolved in water (equivalent to 5.4×106 CFU/mL as determined by standard plate count) by oral gavage daily for 14 days. The carbamazepine group (n=4) was administered carbamazepine at a dose of 75 mg/kg by oral gavage daily. The carbamazepine plus B. bifidum group (n=4) was administered 1 g of B. bifidium probiotics at a concentration of 5.4×106 CFU/mL by oral gavage daily for 14 days and carbamazepine at a dose of 75 mg/kg by oral gavage. The combined effect of carbamazepine and probiotics was assessed by measuring changes in weight, urination, and anxiety. Anxiety was tested using the light/dark transition test as described by Pieróg et al.24
Formulation considerations. For the formulation design, B. bifidum (Nutri-Nature) and carbamazepine purchased from a local pharmaceutical company were used. Prior to formulation, preformulation studies were done on B. bifidum to determine organoleptic properties, pH, flow rate, and bulk density. Color evaluation was done by visual inspection and compared to the standard hexadecimal color code.25 Texture uniformity was assessed by spreading the sample on a white tile to examine its smoothness, firmness, coarseness, and any irregularities. For the susceptibility of B. bifidum to various excipients, such as methylparaben, sodium benzoate, propylparaben, sorbitol, sucrose, citric acid, and carboxymethyl cellulose, the disk diffusion method, recommended by the National Committee for Clinical Laboratory Standards,19 was used.
The probiotic-infused suspension was prepared using the precipitation method.26 Water-insoluble active ingredients were dissolved in a water-miscible organic solvent (glycerin), then added to distilled water under appropriate conditions to ensure proper dispersion and distribution of the active ingredients.
Formulation stability testing. Suspension stability testing involved several evaluations over time, specifically on Days 0, 7, 14, 21, and 28. Organoleptic properties (color, texture, and smell) were assessed as according to Boscolo et al.27 The pH, redispersibility, and sedimentation were evaluated. Bacterial viability was determined using the disk diffusion19 and plate count methods.28
Enumeration of B. bifidum in the probiotic infused suspension. The standard plate count method was used to quantify the bacterial population at regular intervals. One milliliter of the B. bifidum–infused suspension was taken and diluted with 9 mL of distilled water. Serial dilutions were then prepared following the method described by Penfornis et al28 up to 108 dilution factor. Subsequently, 1 mL of the appropriate dilution was spread onto Lactobacillus MRS agar plates. Inoculated plates were incubated at 37 °C for 24 hours.19
Effect of temperature on bacterial viability. The 5 mL sample was acquired and exposed to 3 distinct temperature conditions: 25 °C, 35 °C, and 45 °C. The sample was kept under each temperature for a period of 24 hours. Subsequently, the 5 mL sample treated at each temperature was uniformly distributed onto Lactobacillus MRS agar plates. These plates were subsequently placed in an incubator set at a constant temperature of 37 °C and allowed to incubate for 24 hours.
Stability of carbamazepine in the dosage form. The UV-Vis spectrophotometer (MRC Systems) was used to confirm the stability of carbamazepine in the dosage form. The wavelength for carbamazepine used to confirm stability was 246 nm as according to Kharde et al.29 Baseline correction was performed by measuring the absorbance of a blank sample without carbamazepine to account for any interference from excipients or formulation components. The absorbance of each sample was then measured at the established wavelength using a cuvette. The absorbance data obtained from the samples were analyzed to evaluate the stability of carbamazepine.
Window ledge test. The formulation was subjected to a window ledge test to assess its physical stability during storage. The sample was placed on an elevated surface for an extended period of 1 week. This treatment exposed it to environmental fluctuations, thus providing insights into the formulation’s ability to maintain integrity and quality under real-world storage conditions. The formulation was observed for changes in physical properties, such as separation, precipitation, color, odor, and texture.
Histopathological evaluation. A single mouse was drawn from each experimental group, including the control group, to harvest the liver, brain, and kidney for histopathological evaluation. The collected samples were then fixed in 10% buffered formal saline for at least 24 hours prior to processing. The sections were prepared for staining with hematoxylin and eosin in the histology laboratory at the Department of Biomedical Sciences, Faculty of Medicine, University of Zimbabwe in accordance with steps laid out by Suvarna et al.30
Statistical analysis. Descriptive statistics used included mean, standard deviation, median, and variance calculated for weight and time data. The Kruskal–Wallis test was used to assess the differences in weight and the time spent in different light and dark compartments among the groups. Statistically significant differences were inferred where P<0.05.
Results
Viability testing of bacterial strains in carbamazepine. B. bifidum showed no significant inhibition when grown in the varying carbamazepine concentrations, indicating resistance (Table 1). However, L. acidophilus showed susceptibility to carbamazepine, with varying zone of inhibition values across concentrations. This data influenced the decision to not use L. acidophilus in the probiotic formulation.
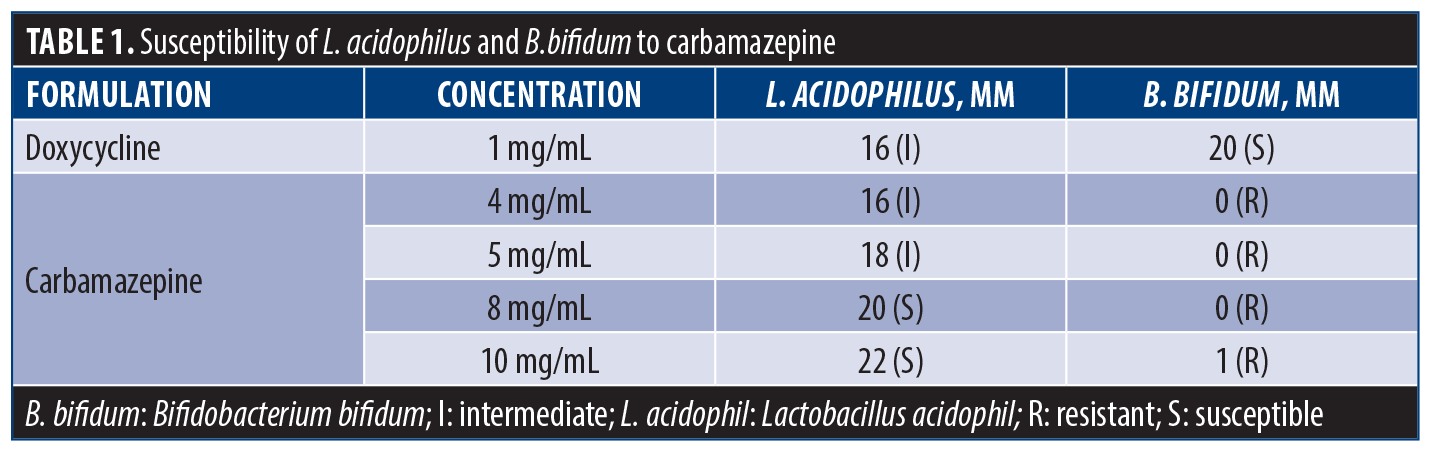
Evaluation of combined effect of probiotic and carbamazepine in animal models. Anxiety behavior. The light-dark test, a behavioral experiment, was used to assess anxiety behavior. In this test, an animal is placed in a box divided into a bright, illuminated compartment and a dark, enclosed compartment. Rodents with higher anxiety levels tend to spend more time in the dark compartment, as they find the bright, open area more aversive.
The light-dark box test revealed significant differences in anxiety levels among rats (P=0.0312). Rats that received B. bifidum only exhibited relatively higher anxiety as compared to those that received carbamazepine only (Figure 1). Interestingly, combined use of carbamazepine and the probiotic resulted in a significant decrease in anxiety-like behavior compared to either treatment alone.
Urination frequency. The frequency of urination was determined by the number of times the bedding of each group was changed in 24 hours. The rats from the probiotic-only group exhibited a similar frequency of urination as those from the control group. However, in the carbamazepine-only group, urination frequency increased. Furthermore, in the carbamazepine plus probiotic group, urination frequency was further heightened. Overall, these findings indicated that the simultaneous use of probiotics and carbamazepine resulted in an increased frequency of urination.
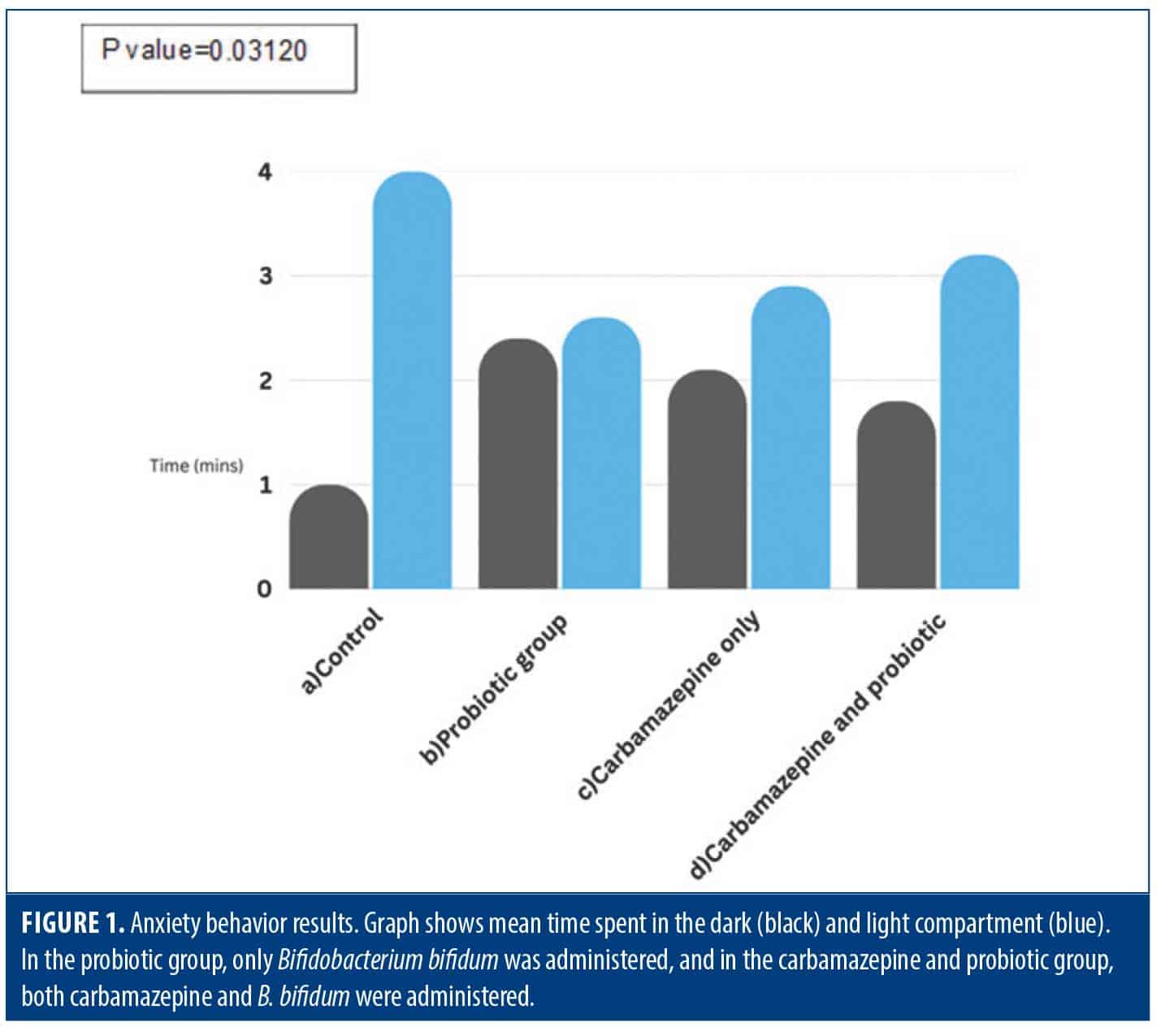
Weight assessment. Body weights were compared before and after the experiment (Figure 2). The data presented in Figure 2 shows the mean weight differences observed across the experimental groups, with a P value of 0.03186. The results indicate that the group receiving probiotics alone and the group receiving carbamazepine plus probiotics experienced a decrease in weight of 2.4 g and 1.8 g, respectively, compared to the control group. In contrast, the control group and the group receiving only carbamazepine exhibited an increase in weight of 3.5 g and 2.7 g, respectively.
Preformulation considerations. Preformulation tests done on B. bifidum prior to formulation met the standard guidelines for color and uniformity.26 B. bifidum had no noticeable odor. B.bifidum culture showed a slightly acidic pH of 4.54. It had a fair flow rate of 12.5 g/s, indicating moderate flowability. The bulk density of 0.8 g/cm3 suggests a reasonable density. Overall, the characteristics of B. bifidum were within acceptable ranges outlined in the British Pharmacopeia.
Growth of B. bifidum in various excipients. Clinical and Laboratory Standards Institute (CLSI) guidelines are not currently available for Bifidiobacterium spp31; thus, interpretation of growth inhibition diameters could not be done. However, testing revealed that methylparaben had the strongest inhibitory effect (18 mm), followed by propylparaben (16 mm), and sorbitol (14 mm). Sucrose showed weak inhibition (1 mm), and sodium benzoate, carboxyl methyl cellulose, and citric acid had minimal effect (0–1 mm). Thus, sodium benzoate was chosen as the preferred preservative for the suspension formulation due to its lack of inhibitory effect on B. bifidum, ensuring its viability.
Suspension formulation. A milky white probiotic-infused carbamazepine suspension was formulated using ingredients that were properly dispersed and within the aqueous medium. Additionally, a separate formulation without probiotics also was prepared to serve as a control during the stability testing.
Stability testing. Viability testing of the probiotic in suspension. The probiotic bacteria, B. bifidum, was able to survive and grow within the formulated suspension. The standard plate count method was used to estimate the number of bacterial colonies, which was found to be approximately 1.3×108 CFU per 5 mL of the suspension. This indicated that the formulation did not hinder the growth and viability of the probiotic bacteria.
Effect of temperature. The suspension maintained a liquid form without any significant thickening at 35 °C. However, the suspension exhibited a thickened consistency at 45 °C, resembling a gum-like substance. Bacterial growth was assessed at varying temperatures. At 25 °C, 3.6×106 CFU were observed, while at 37 °C, the count was 7.7×108 CFU, and at 45 °C, it was 2.3×102 CFU.
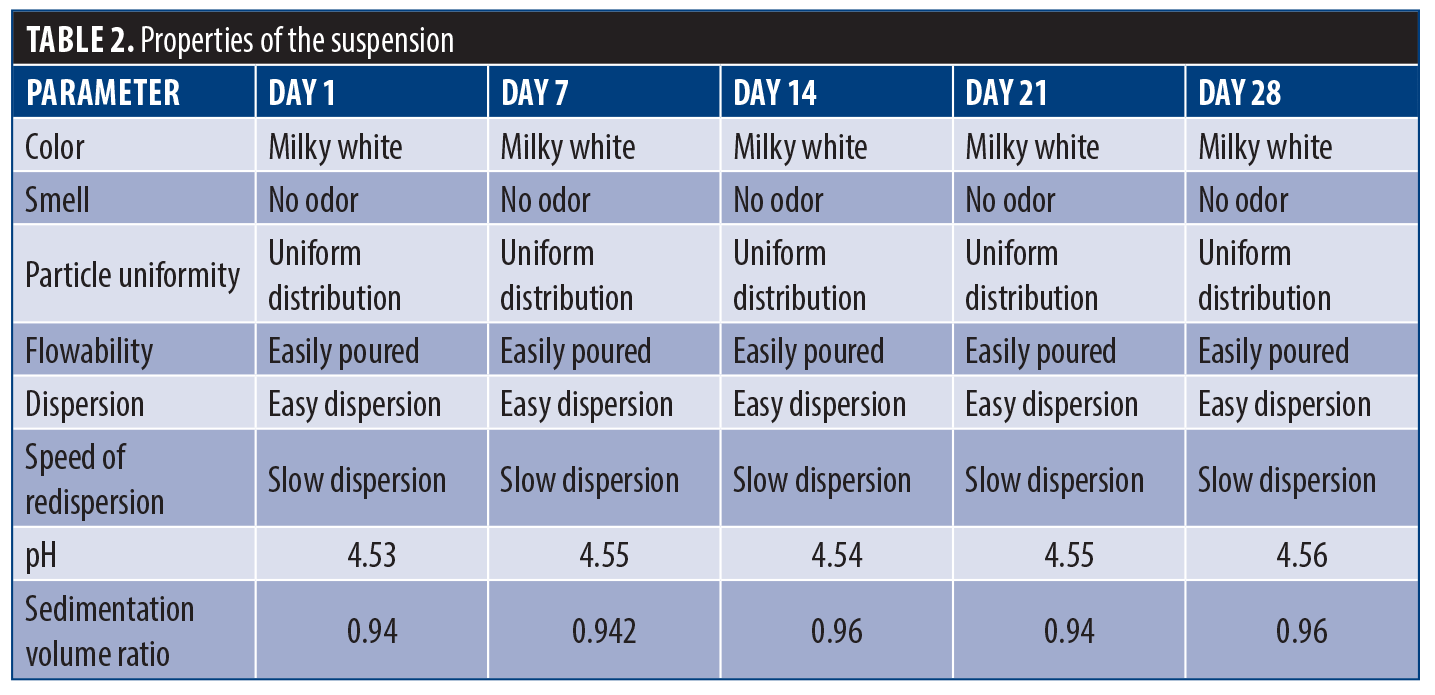
Properties of the suspension. The organoleptic properties (color, odor, particle uniformity, flowability dispersion, and speed of redispersion) of the suspension were evaluated over 28 days (Table 2). The suspension consistently maintained a milky white color with no detectable odor throughout the observation period, indicating that the formulation did not undergo any significant chemical changes that could alter these sensory characteristics. Particle uniformity was also consistently maintained, ensuring a homogeneous mixture. The flowability remained easy, indicating that the suspension did not thicken or become difficult to pour over time. However, while dispersion remained easy, the speed of redispersion was consistently slow, indicating a persistent characteristic that may need further optimization for quicker redispersion. The pH values showed minimal fluctuations, ranging from 4.53 to 4.56 over the 28-day period. The sedimentation volume ratio values ranged from 0.94 to 0.96, indicating a slight decrease in volume due to sedimentation and poor sedimentation stability, with some degree of consistency. These findings suggested that the suspension was generally stable over the 28-day period, meeting most desired parameters, but further optimization may be required to address slow redispersion and sedimentation issues for improved overall stability.
Window ledge test. In summary, the window ledge test, which exposed the suspension to light and environmental conditions, caused the following changes: a color change from milky white to cream, separation of the formulated suspension, and development of a distinct odor. These results indicate that the suspension formulation is unstable and susceptible to physical and/or chemical changes when subjected to light and other environmental factors.
Discussion
The development of a probiotic-infused carbamazepine dosage form is a multifaceted process that involves careful consideration of various factors, including the selection of probiotic strains, formulation methods, excipients, and stability.
The selection of probiotic strains plays a critical role in the development of probiotic-infused carbamazepine dosage forms for epilepsy management. In this study, B. bifidum was chosen over L. acidophilus based on its resistance to carbamazepine, as demonstrated in susceptibility testing according to Gajic et al.21 While the exact mechanisms behind L. acidophilus inhibition by carbamazepine remain unclear, this finding aligns with the unsuitability of L. acidophilus for the probiotic-infused dosage form.
The effects of combined probiotic and carbamazepine administration were evaluated in animal models, focusing on anxiety levels, urination frequency, and weight reduction. The reduction in anxiety-like behavior observed with the combination therapy is consistent with the bidirectional communication between the gut and brain, known as the gut-brain axis, as discussed by Foster and McVey Neufeld.32 The increased urination frequency observed when probiotics were combined with carbamazepine can be attributed to both carbamazepine’s impact on antidiuretic hormone activity and probiotics’ potential role in detoxification processes, as mentioned in studies by Hamer et al33 and Jandhyala et al.34
Weight reduction, particularly when probiotics were combined with carbamazepine, is a significant outcome given carbamazepine’s known adverse effects, such as weight gain and metabolic syndrome. This aligns with the positive impact of probiotics on metabolic health discussed in studies by Elshaghabee et al35 and Depommier et al.36 However, it is essential to note the complexity of probiotic effects on weight, as highlighted by contradictory findings in studies by Sanchez et al,37 and the need for further research to understand specific mechanisms and optimal conditions for achieving weight reduction.
Preformulation studies guided the selection of excipients for the probiotic-infused carbamazepine dosage form. A citric acid buffer was chosen based on the slightly acidic pH of B. bifidum (pH: 4.54) and its need for an acidic environment to thrive. The inhibitory effects of certain excipients, such as parabens and sorbitol, on B. bifidum growth were evaluated, aligning with findings from Crovetto et al38 and Das et al.39 However, sodium benzoate was found to be compatible with B. bifidum growth, consistent with studies by Xiao et al40 and Gerasimidis et al.41
The formulation of a probiotic-infused carbamazepine suspension using the precipitation method resulted in a milky white suspension with well-dispersed ingredients. This method offers the advantage of achieving a uniform distribution of water-insoluble drugs in an aqueous medium, which is crucial for ensuring consistent therapeutic effects. By preparing a control formulation without probiotics, a comparative stability analysis was conducted to understand the impact of probiotics on the formulation’s stability. Previous research, such as the work by Schikarski et al,42 supports the effectiveness of the precipitation method for pharmaceutical suspensions, particularly in achieving homogeneous dispersions of active ingredients. However, the incorporation of probiotics introduces additional considerations for stability due to their potential effects on the chemical environment of the suspension. Comparative studies with control formulations are essential for identifying these impacts, as demonstrated in studies by Baral et al43 that examined the stability of probiotic formulations in various media.
Histopathology is critical in the evaluation of safety and efficacy of a new drug, ruling out potential toxicities, explaining the drug’s mechanism of action, and ultimately determining its suitability for practical clinical use.44 Hyaline casts are made up of protein and may be seen in glomerular disease.45,46 They were seen in the tubules of the kidney samples obtained from the mice from treatment groups as well as those from the control group. Such a finding cannot be attributed to the test compounds since a similar finding was noted in a sample from the control. This can be explained by the fact that hyaline casts may be seen in normal individuals secondary to strenuous exercise, fever, or dehydration.46 The nontoxicity of the test compounds is also supported by the absence of hepatic lesions in the tested samples. Periportal and centrilobular necrosis consistently occur with chemical exposure following metabolic activation of a test compound to toxic metabolites.47 Congestion mainly in the absence of a failing heart is a nonspecific finding. This can happen as a postmortem change due to the passive accumulation of blood.48
Suspensions are suitable for pediatric and geriatric patients who may have difficulty swallowing tablets or capsules. They offer flexible dosing options and can be flavored to improve palatability. Gaikwad et al49 discussed how suspensions provide a versatile dosage form for drugs with solubility issues, leading to better patient adherence and therapeutic outcomes.
Stability studies are essential for determining the shelf life and ensuring the efficacy of pharmaceutical formulations. Prior research, such as that conducted by Schikarski et al42 and Baral et al,43 emphasizes the importance of maintaining organoleptic properties, pH stability, and sedimentation stability in suspensions, as well as providing supportive environments for probiotic viability. Similarly, Hadžiabdić et al50 highlight the negative impact of high temperatures on suspension stability and bacterial viability. The observed stability and bacterial growth patterns in the suspension formulation reinforce the need for lower storage temperatures to ensure efficacy and safety.
The observed stability issues under light and temperature variations align with findings from Gaikwad et al,49 which emphasize the significant influence of environmental factors on suspension stability. The slow redispersion characteristic found in this study is consistent with the challenges highlighted by Malkawi et al51 regarding particle redispersion in suspensions.
Limitations
Despite the promising findings, this study presents several limitations that warrant careful consideration. The findings are predominantly derived from preclinical animal models, which may limit their direct extrapolation to human epilepsy populations. The relatively small sample size may have reduced the statistical power and restricted the generalizability of the results. Although promising synergistic effects between carbamazepine and B. bifidum were observed, the precise mechanisms underlying these interactions were not fully elucidated. Furthermore, long-term safety, pharmacokinetic interactions, and microbiome alterations were not extensively investigated.
Additionally, stability studies were conducted under controlled laboratory conditions, which may not accurately reflect real-world storage environments, particularly in resource-limited settings. Consequently, further large-scale clinical trials and detailed mechanistic investigations are warranted to substantiate the efficacy, safety, and translational applicability of this combined therapeutic approach.
Conclusion
Incorporating probiotics into carbamazepine formulations offers promising benefits, necessitating expanded trials, formulation optimization, economic evaluations, regulatory approvals, and educational campaigns for widespread adoption and impact globally.
Acknowledgements
We would like to acknowledge the invaluable contributions of individuals who supported this work. Special acknowledgement goes to Mr. Kwitiri at the University of Zimbabwe, College of Health Sciences, Animal House Unit for assistance with handling of the animals used in this study.
Author contributions
HM was involved in development of the idea, formulation of the dosage form, and assessment of stability. TH conceptualized the idea, developed the protocol, was involved in all stages of the work, and drafted the manuscript. JC was the laboratory technician who coordinated the experiments conducted throughout the study. SW is the veterinary specialist who assisted with the evaluation of the dosage form in animal models. NDS served as the lead and corresponding author, overseeing the entire project and manuscript preparation.
References
- Olaciregui-Dague K, Weinhold L, Hoppe C, et al. Anti-seizure efficacy and retention rate of carbamazepine is highly variable in randomized controlled trials: a meta-analysis. Epilepsia Open. 2022;7(4):556–569.
- Kubová H, Mares P. Anticonvulsant action of oxcarbazepine, hydroxycarbamazepine, and carbamazepine against metrazol-induced motor seizures in developing rats. Epilepsia. 1993;34(1):188–192.
- Lindefeldt M, Eng A, Darban H, et al. The ketogenic diet influences taxonomic and functional composition of the gut microbiota in children with severe epilepsy. NPJ Biofilms Microbiomes. 2019;5(1):5.
- Xie G, Zhou Q, Qiu C-Z, et al. Ketogenic diet poses a significant effect on imbalanced gut microbiome in infants with refractory epilepsy. World J Gastroenterol. 2017;23(33):6164–6171.
- Liang S, Wu X, Jin F. Gut-brain psychology: rethinking psychology from the microbiota-gut-brain axis. Front Integr Neurosci. 2017;11:33.
- Wei S, Mai Y, Hu L, et al. Altered gut microbiota in temporal lobe epilepsy with anxiety disorders. Front Microbiol. 2023;14:1165787.
- Stopińska K, Radziwoń-Zaleska M, Domitrz I. The microbiota-gut-brain axis as a key to neuropsychiatric disorders: a mini review. J Clin Med. 2021;10(20):4640.
- Mhanna A, Martini N, Hmaydoosh G, et al. The correlation between gut microbiota and both neurotransmitters and mental disorders: a narrative review. Medicine (Baltimore). 2024;105(5):e37114.
- Holzer P, Farzi A. Neuropeptides and the microbiota-gut-brain axis. In: Lyte M, Cryan JF, eds. Microbial Endocrinology: The Microbiota-Gut-Brain Axis in Health and Disease. Advances in Experimental Medicine and Biology, vol. 817. Springer, New York; 2014:195–219.
- Barrett E, Ross RP, O’Toole PW, et al. γ-Aminobutyric acid production by culturable bacteria from the human intestine. J Appl Microbiol.2012;113(2):411–417.
- Mazzoli R, Pessione E. The neuro-endocrinological role of microbial glutamate and GABA signaling. Front Microbiol. 2016;7:1934.
- Chen Y, Xu J, Chen Y. Regulation of neurotransmitters by the gut microbiota and effects on cognition in neurological disorders. Nutrients. 2021;13(6):2099.
- Liu H, Wang J, He T, et al. Butyrate: a double-edged sword for health? Adv Nutr. 2021;12(1):21–29.
- Wen L, Duffy A. Factors influencing the gut microbiota, inflammation, and type 2 diabetes. J Nutr. 2017;147(7):1468S–1475S.
- Purdel C, Ungurianu A, Adam-Dima I, Margină D. Exploring the potential impact of probiotic use on drug metabolism and efficacy. Biomed Pharmacother. 2023;161:114468.
- Wilson ID, Nicholson JK. Gut microbiome interactions with drug metabolism, efficacy, and toxicity. Transl Res. 2017;179:204–222.
- Latif A, Shehzad A, Niazi S, et al. Probiotics: mechanism of action, health benefits and their application in food industries. Front Microbiol. 2023;14:1216674.
- Singh J, Rivenson A, Tomita M, et al. Bifidobacterium longum, a lactic acid-producing intestinal bacterium inhibits colon cancer and modulates the intermediate biomarkers of colon carcinogenesis. Carcinogenesis. 1997;18(4):833–841.
- Kiehlbauch JA, Hannett GE, Salfinger M, et al. Use of the National Committee for Clinical Laboratory Standards guidelines for disk diffusion susceptibility testing in New York state laboratories. J Clin Microbiol. 2000;38(9):3341–3348.
- British Pharmacopoeia Commission. British Pharmacopoeia 2021. London: The Stationery Office. 2020.
- Gajic I, Kabic J, Kekic D, et al. Antimicrobial susceptibility testing: a comprehensive review of currently used methods. Antibiotics (Basel). 2022;11(4):427.
- Fowler JL, Clark WS Jr, Foster JF, Hopkins A. Analyst variation in doing the standard plate count as described in Standard Methods for the Examination of Dairy Products1,2. J Food Prot. 1978;41(1):4–7.
- Njeru PN, Rösch N, Ghadimi D, et al. Identification and characterisation of lactobacilli isolated from Kimere, a spontaneously fermented pearl millet dough from Mbeere, Kenya (East Africa). Benef Microbes. 2010;1(3):243–252.
- Pieróg M, Socała K, Doboszewska U, et al. Effects of new antiseizure drugs on seizure activity and anxiety-like behavior in adult zebrafish. Toxicol Appl Pharmacol. 202115;427:115655.
- Color-hex. Color Hex color codes. Accessed 15 Feb 2023. https://www.color-hex.com/
- British Pharmacopoeia Commission. British Pharmacopoeia 2023. London: The Stationery Office; 2022.
- Boscolo O, Perra F, Salvo L, et al. Formulation and stability study of omeprazole oral liquid suspension for pediatric patients. Hosp Pharm. 2020;55(5):314–322.
- Penfornis P, Pochampally R. Colony forming unit assays. Methods Mol Biol. 2016;1416:
159–169. - Kharde O, Doifode S, Dongaonkar T, Tathe P. Development and validation of UV-visible spectroscopic method for estimation of carbamazepine in bulk and tablet dosage form. World J Pharm Res. 2020;9:84.
- Bancroft JD, Layton C. 10 – The hematoxylins and eosin. In: Survana KS, Layton C, Bancroft JD, eds. Bancroft’s Theory and Practice of Histological Techniques. 8th ed. Elsevier; 2018:126–138.
- Blandino G, Milazzo I, Fazio D. Antibiotic susceptibility of bacterial isolates from probiotic products available in Italy. Microb Ecol Health Dis. 2008;20(4):199–203.
- Foster JA, McVey Neufeld K-A. Gut–brain axis: how the microbiome influences anxiety and depression. Trends Neurosci. 2013;36(5):
305–312. - Hamer HM, Jonkers D, Venema, K, et al. Review article: the role of butyrate on colonic function. Aliment Pharmacol Ther. 2008;27(2):104–119.
- Jandhyala SM, Talukdar R, Subramanyam C, et al. Role of the normal gut microbiota. World J Gastroenterol. 2015;21(29):8787–8803.
- Elshaghabee FM, Rokana,N, Gulhane RD, et al. Bacillus as potential probiotics: status, concerns, and future perspectives. Front Microbiol. 2017;8:1490
- Depofigmmier C, Everard A, Druar C, et al. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med. 2019;25(7):1096–1103.
- Sánchez B, Delgado S, Blanco-Míguez A, et al. Probiotics, gut microbiota, and their influence on host health and disease. Mol Nutr Food Res. 2017;61(1).
- Crovetto SI, Moreno E, Dib AL, et al. Bacterial toxicity testing and antibacterial activity of parabens. Toxicol Environ Chem. 2017;99(5–6):858–868.
- Das SG, Bhattacharjee D, Manna A, et al. Effect of different excipients and packaging materials on commercial preparation of probiotic formulation. Int J Pharm Sci Res. 2014;5(5):1830–1836.
- Xiao N, Ruan S, Mo Q, et al. The effect of sodium benzoate on host health: insight into physiological indexes and gut microbiota. Foods. 2023;12(22):4081.
- Gerasimidis K, Bryden K, Chen X, et al. The impact of food additives, artificial sweeteners and domestic hygiene products on the human gut microbiome and its fibre fermentation capacity. Eur J Nutr. 2020;59(7):3213–3230.
- Schikarski T, Trezenschiok H, Avila M, Peukert W. Impact of solvent properties on the precipitation of active pharmaceutical ingredients. Powder Technol. 2023;415:118032.
- Baral KC, Bajracharya R, Lee SH, Han H-K. Advancements in the pharmaceutical applications of probiotics: dosage forms and formulation technology. Int J Nanomedicine. 2021;12:7535–7556.
- Provenzano E, Driskell OJ, O’Connor D J, et al. The role of the histopathologist in clinical trials, challenges and approaches to tackle them. Histopathol. 2020;76(7):942–949.
- Gonden V, Bhatt H, Jialal I. Renal function tests. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. Updated 27 Jul 2024. Accessed 4 Jan 2025. https://www.ncbi.nlm.nih.gov/books/NBK507821/
- Shikata E, Hattori R, Hara M, Nakayama T. The detection of hyaline casts in patients without renal dysfunction suggests increased plasma BNP. EJIFCC. 2021;32(4):410–420.
- Foster JR, Semino-Beninel G, Melching-Kollmuss S. The cumulative risk assessment of hepatotoxic chemicals: a hepatic histopathology perspective. Toxicol Pathol. 2020;48(3):397–410.
- Grant Maxie M, Miller MA. Chapter 1 – Introduction to the Diagnostic Process. In: Grant Maxie M, ed. Jubb, Kennedy and Palmer’s Pathology of Domestic Animals. 6th ed. Elsevier; 2016:1–15.e1.
- Gaikwad SS, Morales JO, Lande NB, et al. Exploring paediatric oral suspension development: challenges, requirements, and formulation advancements. Int J Pharm. 2024;657:124169.
- Hadžiabdić J, Elezović A, Rahić O, et al. Stability of suspensions: theoretical and practical considerations before compounding. Int J Pharm Compd. 2015;19(1):78–85.
- Malkawi R, Malkawi WI, Al-Mahmoud Y, Tawalbeh J. Current trends on solid dispersions: past, present, and future. Adv Pharmacol Pharm Sci. 2022:5916013.
