Innov Clin Neurosci. 2026;23(1–3):47–53.
by Hamed Zarei, MD*; Mohammadhossein Vazirizadeh-Mahabadi, MD*; Narges Saadatipour, MD; Maryam Pazhooha, MD; Mostafa Hosseini, PhD; and Mahmoud Yousefifard, PhD
*Drs. Zarei and Vazirizadeh-Mahabadi contributed equally to this article.
Drs. Zarei, Vazirizadeh-Mahabadi, Saadatipour, Pazhooha, and Yousefifard are with Physiology Research Center, Iran University of Medical Sciences, Tehran, Iran. Dr. Hosseini is with Department of Epidemiology and Biostatistics, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran.
ABSTRACT: Background: Early detection and outcome prediction of stroke are crucial for enhancing poststroke care. We explored the diagnostic and prognostic potential of bioactive adrenomedullin (bio-ADM) in stroke through a systematic review and meta-analysis. Methods: Medline, Embase, Scopus, and Web of Science were searched for studies measuring bio-ADM in patients with stroke. Diagnostic outcomes were confirmed via imaging, while prognostic outcomes included mortality and modified Rankin Scale (mRS). For meta-analysis, standardized mean differences (SMDs), pooled odds ratios (ORs), and sensitivity/specificity were synthesized. Results: Ten articles were included. Bio-ADM levels were significantly higher in patients with stroke compared to nonstroke control groups (SMD: 1.47; 95% confidence interval [CI]: 0.98–1.97). Subgroup analyses of acute ischemic stroke (SMD: 1.22; 95% CI: 0.34–2.11) and acute hemorrhagic stroke (SMD: 1.67; 95% CI: 1.07–2.26) demonstrated similar findings. Bio-ADM levels within the first 24 hours of a stroke were significantly higher in patients who died compared to those who survived (SMD: 1.35; 95% CI: 1.08–1.61). Pooled data analysis from multivariate models demonstrated no independent association between increased bio-ADM levels within the first 24 hours of admission and mortality (pooled adjusted OR: 1.11; 95% CI: 0.97–1.27; P=0.135). Sensitivity and specificity for mortality prediction were 0.617 (95% CI: 0.516–0.709) and 0.850 (95% CI: 0.772–0.905), respectively. Bio-ADM levels were also higher in patients with mRS score of 3 to 6 compared to those with mRS score of 0 to 2 (SMD: 0.48; 95% CI: 0.16–0.80). Conclusion: Bio-ADM is significantly elevated in both hemorrhagic and ischemic strokes and associated with mortality and poor outcomes, though it is not independently predictive of death. Keywords: Adrenomedullin, stroke, hemorrhagic stroke, peptide hormones, bio-ADM, cerebrovascular disorders, brain infarction, ischemic stroke, early diagnosis, prognosis
Introduction
Globally, stroke ranks among the top causes of death and disability.1 Initiating rehabilitation soon after a stroke can improve functional abilities regained by the patient and minimize lasting disabilities.2 Accurate and timely prediction of outcomes following a stroke is essential for managing the condition effectively and appropriately planning rehabilitation and long-term care.2 Some biomarkers have the potential to help physicians predict patient mortality and unfavorable outcomes, enabling them to personalize rehabilitation programs.3
Adrenomedullin (ADM) is known as a vasoactive peptide and is expressed in a variety of tissues such as blood vessels, skeletal muscles, heart, lungs, and nerve tissues.4 Circulating biologically active ADM (bio-ADM), recognized as a novel and promising biomarker, serves as a key regulatory element in disorders of the nervous system.5 ADM has been demonstrated to act as an intrinsic protective agent against brain damage, with elevated ADM levels observed in both ischemic and hemorrhhagic strokes. Specifically, studies have shown that ADM plays a neuroprotective role in ischemic stroke by modulating nitric oxide synthase and inflammatory mediators. Similarly, it is elevated in traumatic brain injuries, suggesting a potential role in promoting recovery following such events.6 Bio-ADM is also noted for its potent anti-inflammatory properties and its ability to interact with different receptors to maintain vascular integrity and regulate angiogenesis, thus protecting against vascular damage.7 These effects are noted in both acute and chronic cerebral ischemia, and bio-ADM has been shown to support nerve regeneration under pathological conditions, highlighting its crucial role in neurological disorders.5 The expression of bio-ADM is triggered by hypoxia via the activation of the hypoxia-inducible factor-1 (HIF-1) pathway.8
Previous studies have linked elevated serum bio-ADM levels with the severity and outcomes of acute ischemic and hemorrhagic strokes.6,8 Hence, there is no conclusive evidence regarding the prognostic and diagnostic value of bio-ADM in stroke. This systematic review and meta-analysis aims to assess the diagnostic and prognostic utility of bio-ADM in differentiating ischemic and hemorrhagic strokes from stroke mimics, identifying subtypes of strokes, and predicting mortality and unfavorable outcomes.
Methods
Study design. The present systematic review and meta-analysis was designed and conducted adhering to the Preferred Reporting Items for Systematic Review and Meta-Analysis of Diagnostic Test Accuracy Studies (PRISMA-DTA).9 The present study adhered to a registered protocol on the international Prospective Registry of Systematic Reviews (PROSPERO).
This study was designed to assess the diagnostic role of ADM in distinguishing between ischemic and hemorrhagic strokes, as well as differentiating stroke from nonstroke conditions. We also aimed to evaluate the prognostic value of ADM in predicting unfavorable outcomes and mortality in patients with stroke. The PIT framework was structured as follows: P (patient/problem/population) included studies on patients with confirmed or suspected stroke; I (index test) involved bio-ADM; T (target condition) for the diagnostic study part included computed tomography angiography or magnetic resonance angiography, and for the prognostic study part, included mortality and unfavorable outcomes measured by the Glasgow Outcome Scale (GOS), Extended Glasgow Outcome Scale (GOSE), or modified Rankin Scale (mRS).
Search strategy. A systematic search was conducted in Medline (via PubMed), Embase, Scopus, and Web of Science databases up to November 10, 2023. The keywords for the search strategy were derived from MeSH and Emtree terms associated with “adrenomedullin” and “stroke,” along with their synonyms. The search strategy was further refined through consultation with an expert in the field. Details of the queries are presented in Appendix 1. Additionally, gray literature, citation tracking, and reference lists from included studies were examined to identify any potentially missing studies.
Eligibility criteria. Observational studies assessing the mortality and unfavorable outcomes of patients with stroke who underwent serum bio-ADM assessment were included in this study. This also encompassed studies comparing the serum levels of bio-ADM in patients with stroke vs patients without stroke and ischemic vs hemorrhagic strokes. Excluded were retracted articles, letters, reviews, abstracts, studies employing other biomarkers, studies lacking the desired outcome measures, studies using pro-ADM as the index test, and studies focusing on the therapeutic role of bio-ADM.
Screening and data collection. The obtained records from systematic search were imported into Endnote X9 software (Thomson Reuters). After omitting the duplicate records, 2 independent reviewers screened the title and abstract of studies and selected potentially relevant articles. Full text of the records were thoroughly reviewed, and related articles were selected based on eligibility criteria. Any disagreements were resolved by a third reviewer in any stage of the screening.
Two separate Excel sheets (prognostic and diagnostic) were designed for summarizing the data of the included studies. At all stages, the 2 reviewers were blinded to each other’s decisions. The extracted data included information about study design and methodology, participant demographics and baseline characteristics, numbers of patients in both the outcome and nonoutcome groups, and serum levels of ADM in these groups. Additionally, the ADM cutoff value, true positives (TPs), false positives (FPs), true negatives (TNs), false negatives (FNs), odds ratios (ORs), relative risks (RRs), and hazard ratios (HRs) were summarized into data sheets.
Risk of bias assessment and the level of evidence. Risk of bias assessment was independently carried out by 2 independent reviewers using the Quality Assessment tool for Diagnostic Accuracy Studies (QUADAS-2) checklist. The 2 review authors resolved any disagreements by discussion. Additionally, Grading of Recommendations Assessment, Development and Evaluation (GRADE) was used to assess the level of evidence.10
Statistical analysis. All statistical analyses were conducted using STATA 17.0 (StataCorp LLC). The data we planned to synthesize were of 3 types: standardized mean differences (SMDs) of ADM serum levels between the outcome and nonoutcome groups (using Hedges’ g, which does not require equalization of units between studies), diagnostic/prognostic accuracy data (TP, TN, FP, and FN), and summary statistics (OR, RR, or HR). The minimum number of studies required for synthesis was 3 for performing pooled analyses. Due to methodological heterogeneity among studies, a random-effects model was used. In cases where meta-analysis was not applicable due to data scarcity, we qualitatively reported a summary of the studies’ findings.
For each outcome with a sufficient number of studies, subgroup analysis was planned based on the type of stroke. Subsequently, meta-regressions were conducted to search for potential sources of heterogeneity. Statistical heterogeneity was assessed using the I2 statistic and the χ2 test, with P<0.1 or I2>50% indicating significant heterogeneity. Publication bias was evaluated using funnel plots and modified Eager’s test proposed by proposed by Doleman et al.11
Results
Search results. The systematic search yielded 697 unduplicated records. After primary screening, a detailed full-text review of 37 studies was conducted. Five studies that assessed the prognostic or diagnostic role of pro-ADM were excluded from the full-text review.12–16 Additionally, 3 studies were not included in the quantitative data synthesis; one used GOSE as the indicator of unfavorable outcome,17 another lacked sufficient data for meta-analysis,18 and the third involved serum sampling upon admission to a rehabilitation center after hospital discharge, which could not be pooled with acute measurements by our studies.2 Ultimately, 10 articles2,6,8,17–23 were included in this systematic review, with 7 of these being incorporated into the meta-analysis.6,8,19–23 Manual searches on Google, Google Scholar, and efforts in reference searching and citation tracking did not identify any additional studies (Figure 1).
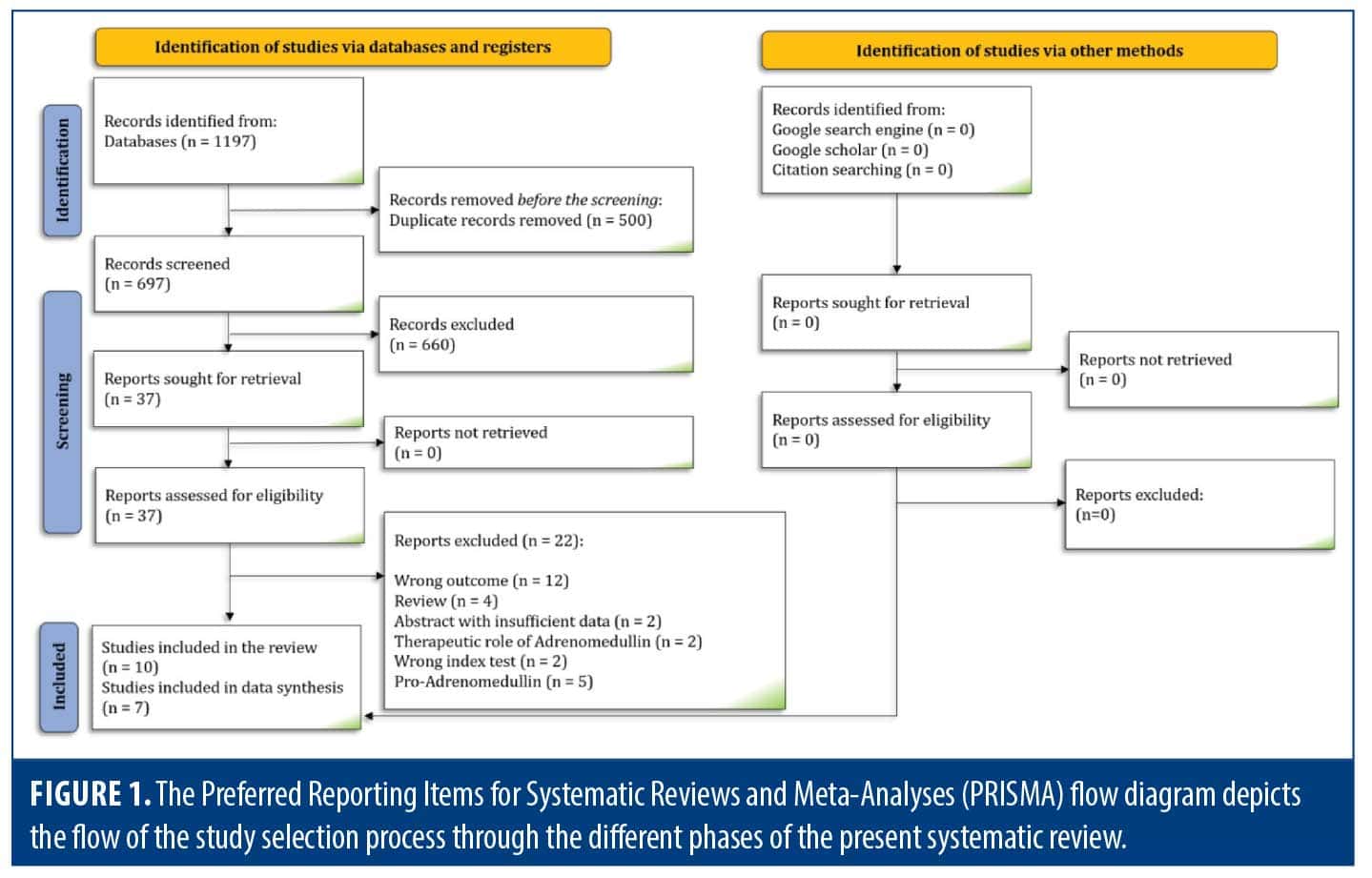
Characteristics of the included studies. Of the 10 studies included, 9 articles investigated the role of serum bio-ADM in stroke diagnosis, and 7 conducted the comparison between the 2 groups of patients with positive or negative radiologic findings as the reference standard. There was only 1 study comparing bio-ADM levels between hemorrhagic and ischemic stroke. One study utilized data from 2 population-based cohort studies with long follow-ups to calculate the HR for the association of bio-ADM levels in patients without stroke with the risk of future stroke incidence. Differences in population and methodology prevented the inclusion of this study in the meta-analysis.
Prognostic performance of bio-ADM in predicting 3- to 6-month mortality was assessed in 3 studies. Also, prognostic yield of bio-ADM to predict the neurological functional outcome was evaluated in 4 studies based on mRS. One study reported the functional outcome based on GOSE (Table 1). All included studies employed immunoassay techniques to detect bio-ADM, with ELISA being the most commonly used platform, utilized in 4 out of 10 studies (Supplementary Table 1).
Risk of bias assessment. Patient selection was not random or consecutive in 8 studies and unclear in 1 study. Regarding the index test, either the interpretation of bio-ADM level was conducted without the knowledge of reference standard or the threshold of bio-ADM was not prespecified, causing an unclear answer in 4 studies in this domain. Additionally, it was not clear if the interpretation of reference standard was done without the knowledge of bio-ADM level in 7 studies. All of the included papers had some concerns regarding the risk of bias assessment based on QUADAS-2 tool. Details are provided in Table 2.
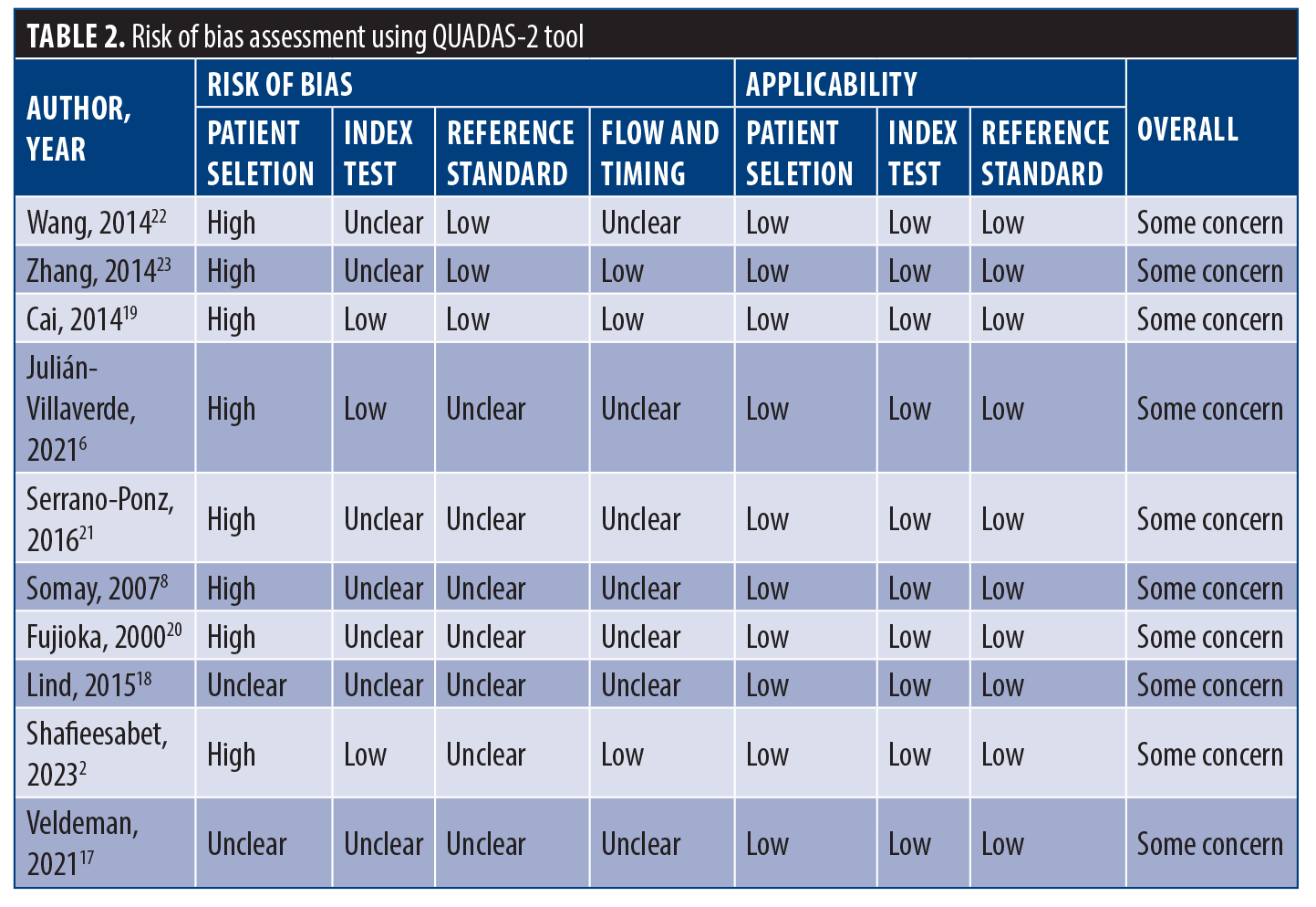
Diagnostic role of bio-ADM in ischemic and hemorrhagic stroke. Pooled data analysis revealed that bio-ADM level was significantly higher in patients diagnosed with stroke compared to nonstroke control groups (SMD: 1.47; 95% CI: 0.98–1.97; I2: 90.77%). Subgroup analyses of acute ischemic stroke (SMD: 1.22; 95% CI: 0.34–2.11; I2: 92.48%) and acute hemorrhagic stroke (SMD: 1.67; 95% CI: 1.07–2.26; I2: 88.18%) demonstrated similar trends (Figure 2). Meta-regression demonstrated no significant difference of effect sizes between ischemic and hemorrhagic strokes (meta-regression coefficient: 0.42; 95% CI: –0.61 to 1.45; P=0.422). Sensitivity analysis restricted to studies with a stroke-to-sampling interval of less than 24 hours revealed consistent results (Supplementary Figure 1).
Prognostic role of bio-ADM in ischemic and hemorrhagic stroke. Pooled data analysis demonstrated that bio-ADM levels measured within the first 24 hours of a stroke were significantly higher in patients who died compared to those who survived (SMD: 1.35; 95% CI: 1.08–1.61; P<0.001; I2: 0.00%; Supplementary Figure 2). Subgroup analysis and meta-regression based on the stroke type were not feasible due to an insufficient number of studies.
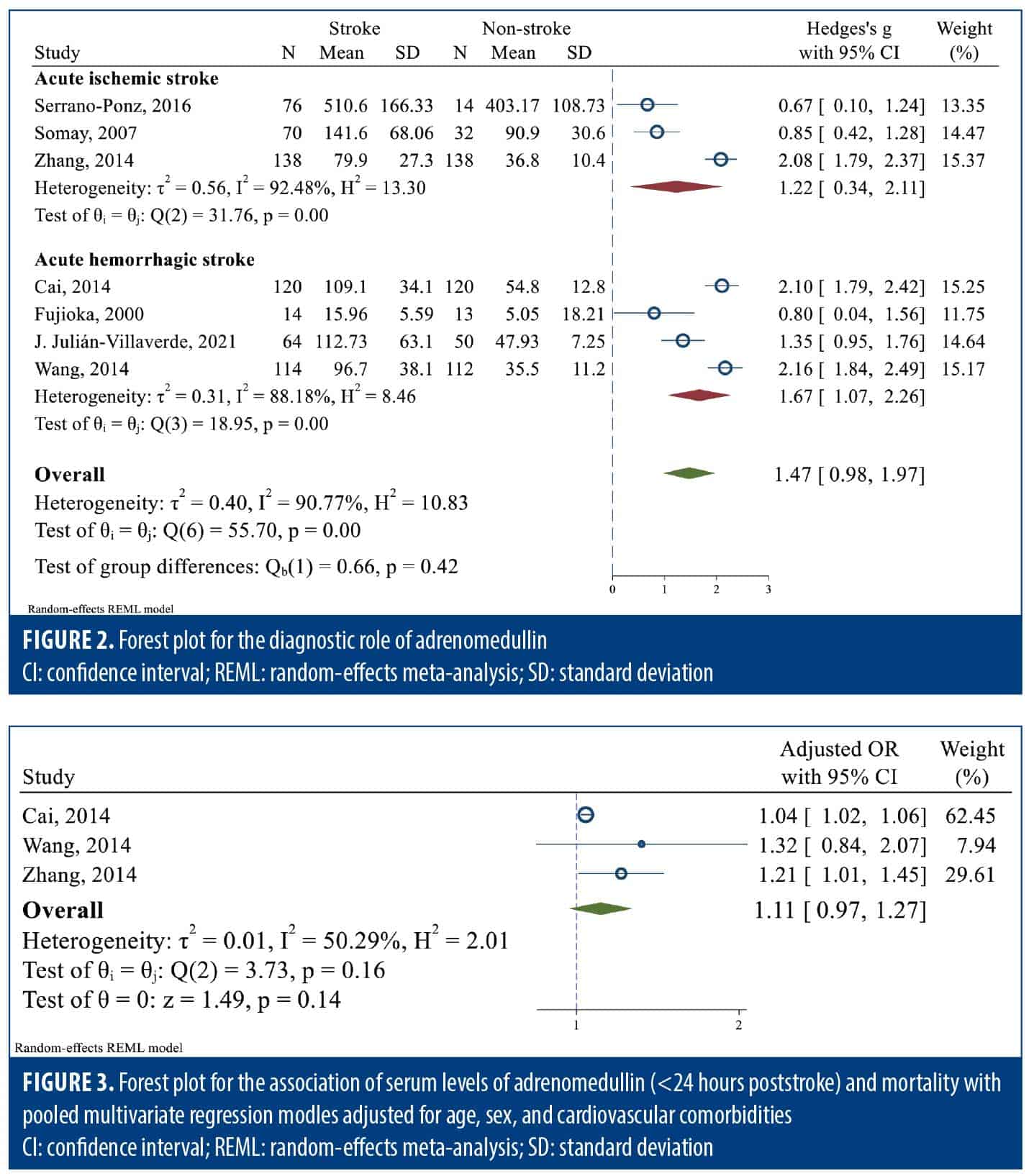
ORs of mortality retrieved from multivariate analyses adjusted for age, sex, and cardiovascular comorbidities were reported in 3 studies. Pooled data analysis demonstrated no independent association between increased bio-ADM levels within the first 24 hours of admission and mortality (pooled adjusted OR: 1.11; 95% CI: 0.97–1.27; P=0.135; I2: 50.29%; Figure 3). In other words, although the mortality of stroke seems to be associated with the level of bio-ADM, this association was not confirmed to be independent in multivariate regression analyses adjusting for age, sex, and cardiovascular comorbidities. We also assessed the prognostic yield of bio-ADM for mortality, with cutoff points ranging from 70 to 122 pg/mL. For predicting mortality among patients with stroke, bio-ADM exhibited an area under the curve, sensitivity, and specificity of 0.752 (95% CI: 0.691–0.804), 0.617 (95% CI: 0.516–0.709), and 0.850 (95% CI: 0.772–0.905), respectively (Supplementary Figure 3).
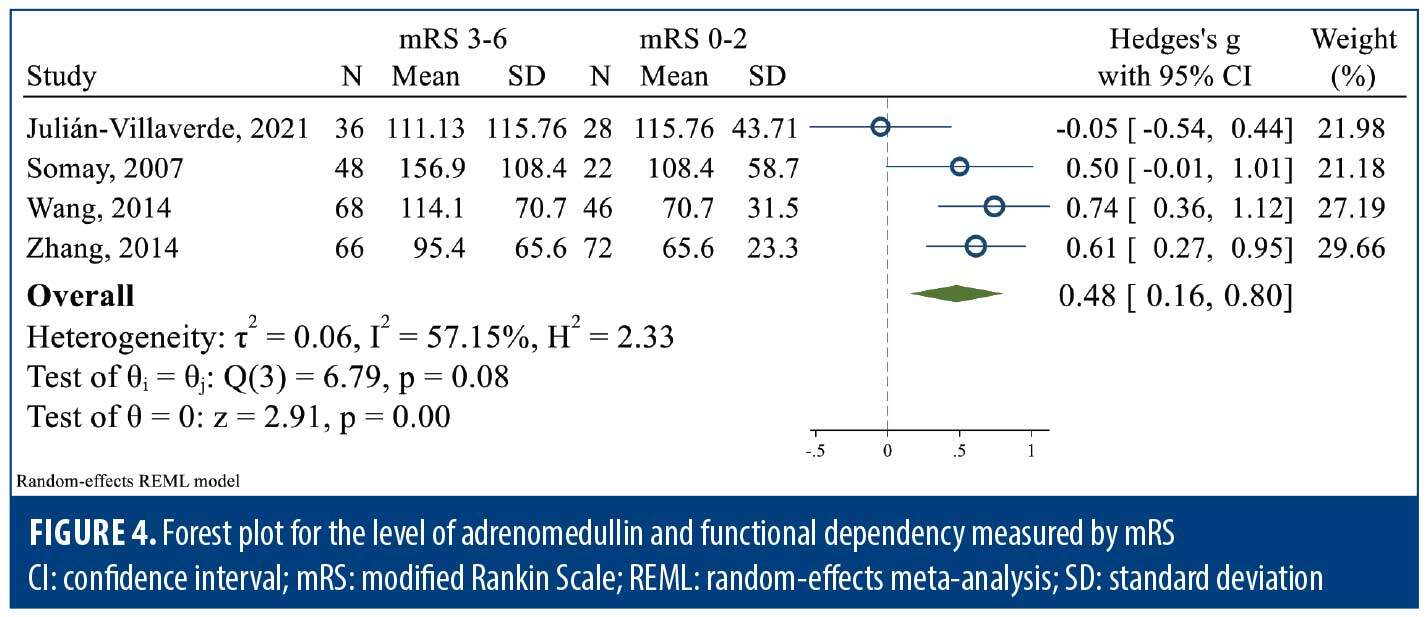
Functional dependency was assessed in 4 studies based on mRS scale. Four studies were included in the pooled data analysis, and mRS score of 3 to 6 was considered an unfavorable outcome. The level of bio-ADM was significantly higher in the unfavorable group compared to the favorable group (SMD: 0.48; 95% CI: 0.16–0.80; I2: 57.15%) (Figure 4). Sensitivity analysis limited to studies measuring bio-ADM within the first 24 hours poststroke resulted in the exclusion of 1 study and revealed no significant difference between the unfavorable and favorable outcome groups (SMD: 0.86; 95% CI: –0.05 to 1.78; I2: 92.97%; Supplementary Figure 4).
Publication bias. Publication bias was assessed for diagnostic and prognostic ability of bio-ADM in stroke. There was no evidence of publication bias for distinguishing stroke from nonstroke, mortality, and functional independence (Supplementary Figure 5).
Level of evidence. The level of evidence regarding the diagnostic and prognostic performance of bio-ADM for detecting stroke and predicting mortality and unfavorable outcomes was determined to be low due to being rated down for the presence of risk of bias and inconsistency (Table 3).

Discussion
This systematic review and meta-analysis investigated the role of bio-ADM in the diagnosis of stroke and in predicting mortality and unfavorable outcomes in patients with stroke. Our results demonstrated that bio-ADM levels werere higher in patients with stroke, both in ischemic and hemorrhagic subtypes. Additionally, the level of bio-ADM can be considered as a predictor of unfavorable functional outcome in patients with stroke. These findings were seen in both ischemic and hemorrhagic stroke.
A possible explanation for the weaker association between bio‑ADM levels and functional outcome observed in the sensitivity analysis restricted to studies with blood sampling within 24 hours is that bio‑ADM measured later in the clinical course may better capture the brain’s post‑treatment ischemic status. Early measurements are likely dominated by the acute vascular and inflammatory response to the initial insult, whereas later sampling can reflect the combined effects of reperfusion, evolving microvascular dysfunction, and secondary neuroinflammation.5,8,24 These processes could result in more stable and integrated bio‑ADM levels that correlate more strongly with long‑term functional outcomes.
The study by Shafieesabet et al2 was the only one that compared serum bio-ADM levels between patients with hemorrhagic and ischemic stroke; the difference was not significant. As only this study met our inclusion criteria, a meta‑analysis was not feasible. Several factors may explain this finding. Sampling occurred at a mean of 25.8±22.1 days poststroke, potentially missing early peak differences. Both subtypes share key mechanisms—cerebral hypoperfusion, blood–brain barrier disruption, vascular stress, and inflammation—that stimulate bio‑ADM release via the HIF‑1 pathway,5,8 likely producing similar elevations. Unequal distribution of comorbidities known to raise bio‑ADM (eg, cardiovascular disease, diabetes, atrial fibrillation, chronic kidney disease)2 and assay or pre‑analytical variability can also mask differences. Together, these factors could explain why Shafieesabet et al did not find significant subtype‑related differences.
Regarding stroke prediction using bio-ADM measurements years before presentation, Lind et al18 investigated the association between bio-ADM levels and the incidence of ischemic stroke in 2 large elderly cohorts (n=1,697). Bio-ADM levels were found to be associated with the incidence of ischemic stroke when adjusted for age (HR: 1.33; 95% CI: 1.05–1.69; P=0.018) and multiple cardiovascular risk factors (HR: 1.39; 95% CI: 1.06–1.82; P=0.018). Veldman et al17 reported an unfavorable outcome based on the GOSE scale, which aligned with the current meta-analysis using the mRS. They found that a long-term (12-month) unfavorable outcome was associated with higher plasma bio-ADM levels during the acute phase (P<0.001).
Bio-ADM is recognized as a key factor in various brain injury conditions, including hemorrhagic and ischemic strokes, as well as traumatic brain injuries.22,23 In the context of brain injuries, bio-ADM may act as a regulator that aids in nerve regeneration. Consequently, bio-ADM is an important biochemical substance in the pathology of nervous system disorders.5,8 Previous research has not firmly established the relationship between bio-ADM levels and the prognosis of patients with stroke. It is anticipated that higher bio-ADM levels would be found in patients with a poorer prognosis, as it is believed to function as a protective peptide. However, recent studies indicate that bio-ADM may have varied effects on the brain and central nervous system, suggesting that its influence on brain injury might stem from the combined actions within these regions.5,6
Pooled data analysis resulted in no independent association between increased bio-ADM levels within the first 24 hours of admission and mortality. However, some research, such as the study by Wang et al,22 showed that there was a significant correlation between mortality in the first 3 months and bio-ADM. In a study by Shafieesabet et al,2 higher levels of bio-ADM were detected in patients with diabetes, atrial fibrillation, cardiovascular disease, and chronic kidney disease. Therefore, cardiovascular diseases could be considered a potential confounding factor in this matter. This could explain why, when mortality rates were adjusted for age, sex, and cardiovascular comorbidities, the independent association between bio-ADM levels and mortality became insignificant.
Biomarkers have been the subject of ongoing research as crucial tools for predicting outcomes in critically ill individuals with the aim of enhancing resource allocation and aiding in clinical decision-making. The process of developing biomarkers has proven to be quite challenging in the context of stroke, as it plays a crucial role in determining both the diagnosis and prognosis of stroke, as well as in predicting the extent of functional recovery following stroke.25 This highlights the significance of utilizing rapid and easily applicable tests, such as bio-ADM, as they could provide valuable aid in the prediction of patient outcomes and formulation of optimal rehabilitation strategies essential for the establishment of a comprehensive poststroke recovery program. The integration of biomarkers with clinical expertise offers valuable insights that are essential in the customization of a patient’s rehabilitation plan.26 This emphasizes the importance of combining biomarkers with clinical judgment to form an optimized treatment plan based on patients’ clinical status, ultimately leading to a more effective recovery process.27
Despite the acceptable sensitivity and specificity of bio-ADM, numerous studies investigated pro-ADM instead, as a consequence of difficulty of measuring bio-ADM in peripheral blood due to its complex biochemical form and rapid clearance from circulation.12–15 Pro-ADM can be located in a variety of organs and tissues, including the endothelium, where it is known to contribute to vasodilation, vasoprotection, and angiogenesis, contributing to its potential therapeutic applications.28,29 Studies have demonstrated a significant association between the increased level of pro-ADM and SAH15 and ischemic12 and hemorrhagic stroke, as well as mortality of stroke13 and acceleration of the atherosclerotic process.14
Limitations. The present study highlights the diagnostic and prognostic value of bio-ADM in hemorrhagic and ischemic stroke. However, our conclusions are primarily limited to unfavorable outcomes assessed by the mRS, as there were insufficient studies reporting outcomes using GOS and GOSE. Moreover, the available studies exhibited substantial methodological heterogeneity, which complicated deeper analyses. Another notable limitation is the fragmented nature of the literature; many researchers have focused on other molecules fragmented concurrently with bio-ADM from their precursors, such as pro-ADM, resulting in scattered and inconsistent data in this field.
The reason we focused exclusively on bio-ADM was due to our predefined eligibility criteria and study protocol. Furthermore, the studies on pro-ADM examined different outcomes, and the number of such studies was insufficient to perform a meta-analysis on any specific outcome.
Due to the relatively small number of eligible studies, subgroup analyses and meta-regressions were limited to stroke type (hemorrhagic vs ischemic) for assessing the diagnostic utility of bio-ADM in differentiating stroke from nonstroke conditions. Even within these subgroups, statistical heterogeneity remained high, likely reflecting differences in sampling timing, assay platforms (although all used immunoassay methods), and pre-analytical factors, as well as the inherent lability of bio-ADM itself. Additionally, the number of studies included in each meta-analysis was insufficient to support further subgroup analyses or meta-regressions based on assay characteristics.
Despite these limitations, this systematic review and meta-analysis comprehensively evaluated the available evidence on bio-ADM in stroke, underscoring its potential utility in patient management and guiding tailored treatment strategies based on expected outcomes.
Conclusion
Bio-ADM is an important biomarker that is enhanced in patients with both hemorrhagic and ischemic stroke. The serum level of bio-ADM is a strong predictor of unfavorable outcomes assessed with mRS in patients with stroke. The level of bio-ADM was found to be higher in patients with stroke who died; however, there was no independent association between this biomarker and mortality. This highlights the importance of bio-ADM in pathophysiology of stroke and its potential clinical use. However, more studies are needed in order to evaluate the impact of bio-ADM on other outcomes in patients with stroke and to find the possible confounders for the existing knowledge on bio-ADM.
Supplementary Materials
To access Supplementary Materials, please visit https://innovationscns.com/wp-content/uploads/Yousefifard_Supplementary_Materials.pdf.
Data availability statement
The dataset generated and analyzed during the current study is available from the corresponding author upon reasonable request.
Author contributions
Study design: MY, MH; Data gathering: HZ, NS, MP; Analysis: HZ, MY; Interpretation: HZ, MV, MY, MH; Drafting: SV, HZ; Revision: all authors; Reading and approving the final manuscript: all authors.
References
- Katan M, Luft A. Global burden of stroke. Semin Neurol. 2018;38(2):208–211.
- Shafieesabet A, Jauert N, Hartmann O, et al. Plasma bioactive adrenomedullin predicts outcome after acute stroke in early rehabilitation. Sci Rep. 2023;13(1):4873.
- Stinear CM, Smith M-C, Byblow WD. Prediction tools for stroke rehabilitation. Stroke. 2019;50(11):3314–3322.
- Shah RV, Truong QA, Gaggin HK, et al. Mid-regional pro-atrial natriuretic peptide and pro-adrenomedullin testing for the diagnostic and prognostic evaluation of patients with acute dyspnoea. Eur Heart J. 2012;33(17):
2197–2205. - Li FJ, Zheng SR, Wang DM. Adrenomedullin: an important participant in neurological diseases. Neural Regen Res. 2020;15(7):1199–1207.
- Julián-Villaverde FJ, Ochoa-Callejero L, Siles E, et al. Adrenomedullin is a diagnostic and prognostic biomarker for acute intracerebral hemorrhage. Curr Issues Mol Biol. 2021;43(1):324–334.
- Ashizuka S, Kita T, Inatsu H, Kitamura K. Adrenomedullin: a novel therapeutic for the treatment of inflammatory bowel disease. Biomedicines. 2021;9(8):1068.
- Somay G, Halac GU, Uslu E, Aydin S. Plasma adrenomedullin in acute ischemic stroke. Neurosciences (Riyadh). 2007;12(4):351–353.
- McInnes MDF, Moher D, Thombs BD, et al. Preferred Reporting Items for a Systematic Review and Meta-Analysis of Diagnostic Test Accuracy Studies: the PRISMA-DTA statement. JAMA. 2018;319(4):388–396.
- Schünemann HJ, Mustafa RA, Brozek J, et al. GRADE guidelines: 21 part 2. Test accuracy: inconsistency, imprecision, publication bias, and other domains for rating the certainty of evidence and presenting it in evidence profiles and summary of findings tables. J Clin Epidemiol. 2020;122:142–152.
- Doleman B, Freeman SC, Lund JN, et al. Funnel plots may show asymmetry in the absence of publication bias with continuous outcomes dependent on baseline risk: presentation of a new publication bias test. Res Synth Methods. 2020;11(4):522–534.
- Seifert-Held T, Pekar T, Gattringer T, et al. Plasma midregional pro-adrenomedullin improves prediction of functional outcome in ischemic stroke. PLoS One. 2013;8(7):e68768.
- Funke-Kaiser A, Havulinna AS, Zeller T, et al. Predictive value of midregional pro-adrenomedullin compared to natriuretic peptides for incident cardiovascular disease and heart failure in the population-based FINRISK 1997 cohort. Ann Med. 2014;46(3):155–162.
- Kawano T, Miyashita K, Takeuchi M, et al. Blood biomarkers associated with neurological deterioration in patients with acute penetrating artery territory infarction: a multicenter prospective observational study. Int J Stroke. 2018;13(2):207–216.
- Gracia Arnillas MP, Alvarez-Lerma F, Masclans JR, et al. Impact of adrenomedullin levels on clinical risk stratification and outcome in subarachnoid haemorrhage. Eur J Clin Invest. 2020;50(11):e13318.
- Ishiyama H, Tanaka T, Saito S, et al. Plasma mid-regional pro-adrenomedullin: a biomarker of the ischemic penumbra in hyperacute stroke. Brain Pathol. 2023;33(2):e13110.
- Veldeman M, Dogan R, Weiss M, et al. Levels of bioactive adrenomedullin in plasma and cerebrospinal fluid in relation to delayed cerebral ischemia in patients after aneurysmal subarachnoid hemorrhage: a prospective observational study. J Neurol Sci. 2021;427:117533.
- Lind L, Siegbahn A, Lindahl B, et al. Discovery of new risk markers for ischemic stroke using a novel targeted proteomics chip. Stroke. 2015;46(12):3340–3347.
- Cai JY, Chen XD, Ba HJ, et al. Identification of plasma adrenomedullin as a possible prognostic biomarker for aneurysmal subarachnoid hemorrhage. Peptides. 2014;59:9–13.
- Fujioka M, Nishio K, Sakaki T, et al. Adrenomedullin in patients with cerebral vasospasm after aneurysmal subarachnoid hemorrhage. Stroke. 2000;31(12):3079–3083.
- Serrano-Ponz M, Rodrigo-Gasqué C, Siles E, et al. Temporal profiles of blood pressure, circulating nitric oxide, and adrenomedullin as predictors of clinical outcome in acute ischemic stroke patients. Mol Med Rep. 2016;13(5):3724–3734.
- Wang CL, Lin HY, Xu JW, et al. Blood levels of adrenomedullin on admission predict outcomes after acute intracerebral hemorrhage. Peptides. 2014;54:27–32.
- Zhang H, Tang B, Yin CG, et al. Plasma adrenomedullin levels are associated with long-term outcomes of acute ischemic stroke. Peptides. 2014;52:44–48.
- Igarashi K, Sakurai T, Kamiyoshi A, et al. Pathophysiological roles of adrenomedullin-RAMP2 system in acute and chronic cerebral ischemia. Peptides. 2014;62:21–31.
- Burke HB. Predicting clinical outcomes using molecular biomarkers. Biomark Cancer. 2016;8:89–99.
- Gandolfi M, Smania N, Vella A, et al. Assessed and emerging biomarkers in stroke and training-mediated stroke recovery: state of the art. Neural Plast. 2017;2017:1389475.
- Stinear CM. Prediction of motor recovery after stroke: advances in biomarkers. Lancet Neurol. 2017;16(10):826–836.
- Gumusel B CJ, Hyman A, Lippton H. Adrenotensin: an ADM gene product with the opposite effects of ADM. Life Sci. 1995;57(8): PL87–90.
- Sugo S, Minamino N, Shoji H, et al. Production and secretion of adrenomedullin from vascular smooth muscle cells: augmented production by tumor necrosis factor-alpha. Biochem Biophys Res Commun. 1994;203(1):719–726.
