Innov Clin Neurosci. 2026;23(1–3):17–22.
by Kenny Nguyen, PharmD; Tammie Lee Demler, PharmD, MBA, BCGP, BCPP;
and Eileen Trigoboff, PMHCNS-BC, DNS, DABFN
Drs. Nguyen and Demler are with The New York State Office of Mental Health at Buffalo Psychiatric Center, Buffalo, New York. Drs. Demler and Trigoboff are with State University of New York at Buffalo School of Pharmacy and Pharmaceutical Sciences, Department of Pharmacy Practice, Buffalo, New York. Dr. Demler is additionally with State University of New York at Buffalo School of Medicine, Department of Psychiatry, Buffalo, New York.
FUNDING: No funding was provided for this article.
DISCLOSURES: The authors have no relevant conflicts of interest.
ABSTRACT: Purpose: Many psychiatric inpatients are prescribed complex medication regimens that include both psychiatric and medical drugs with high anticholinergic (ACH) activity. The cumulative ACH burden (ACB), arising from the pharmacodynamic additive effects of concomitant medication use, has been linked to a range of adverse effects. These include both short-term effects and long-term consequences. Our study aims to determine the degree of ACB in a psychiatric inpatient hospital using current measurement assessment tools. Methods: This retrospective, observational study collected medication regimens and demographic data from the electronic health records of psychiatric inpatients. We included 250 adults aged 18 years or older who were institutionalized in a psychiatric hospital. The ACB was assessed using the ACH toxicity score (ATS), with data collected at two distinct time points, one in the spring and another in the winter, from different calendar years. The total ACB for both psychiatric and medical medications was compared across the patient population, analyzing the potential seasonal variation in the cumulative burden. Results: The degree of total ACB was not statistically significantly different (P=0.526) between the seasonal cohorts and remained consistent with an average total ATS of 8. An ATS score of 5 or higher is assumed to be clinically significant, with 75% of patients (93/124) and 78.6% (99/126) having shown significant burden in the winter and spring cohorts, respectively. Conclusion: The prescribing of ACH medication did not significantly vary seasonally, however, most patients observed showed significant and consistent levels of ACB. It is recommended that clinicians should consider monitoring ACB in their prescribed regimens and consider pharmacologic strategies to reduce ACB. Risk reduction strategies include discontinuation of nonessential ACH medications or consideration of therapeutic interchange with an alternative agent with a lesser ACH profile. The deprescribing of long-term ACH medications for individuals who are otherwise clinically stable may improve both clinical outcomes and quality of life. Keywords: Anticholinergic burden, Beers criteria, anticholinergic adverse effects
Introduction
Anticholinergic (ACH) burden (ACB) represents a critical, yet often overlooked, pharmacological consideration. The risk of ACB is magnified by prescribing trends that utilize polypharmacy, which is a well-recognized, increasingly common trend seen in the aging population and is associated with multimorbidity.1–3 The surge in medication use may reflect beneficial advances in chronic disease management and drug research but also results in detrimental consequences that include a greater likelihood of drug interactions, adverse effects, and a state of being overmedicated. The proportion of older adults prescribed ACH medications increased from 20.7% in 1995 to 23.7% in 2010, with high ACH exposure rising from 7.3% to 9.9% during the same time period.4,5 Antidepressants, some of which are highly ACH, have nearly doubled in prescribing in developed countries between 2000 and 2017.6 ACH medications have a broad range of indications and are likely to continue to be widely utilized.
There is extensive research on the impact of prescribing and the resulting impact of high ACB in at-risk populations. Peripheral and central adverse effects have been noted in the general population; however, they are not as frequent or severe as those observed in older adults and patients with psychiatric disorders, and thus less researched. Up to 50% of older adults are prescribed at least one ACH medication, with up to roughly 12% exposed to clinically significant burden.7,8 Peripheral effects of ACB include constipation, dry mouth, tachycardia, and urinary retention, and central side effects include agitation, confusion, delirium, and cognitive impairment. Elderly patients are more vulnerable to significant ACB due to age-related changes in pharmacokinetic and pharmacodynamic parameters, in addition to cognitive and physical decline. In individuals with psychotic disorders, polypharmacy often includes the use of medications having highly ACH properties, and the total number of medications only increases with illness duration. Since psychotic disorders may be chronic and lifelong, many patients on psychotropic medications are at risk of experiencing iatrogenic, central cholinergic dysfunction.9–11 A study evaluating the degree of ACB in geriatric inpatients revealed that most patients with psychiatric illnesses had a higher likelihood of receiving ACH medications and were overall exposed to significant burden.12 Given the heightened susceptibility of geriatric patients to ACB and the significant exposure often observed in psychiatric patient populations, these two groups should be prioritized when addressing and mitigating the risks associated with ACB.
According to the Beers criteria established by the American Geriatrics Society, ACH medications are recognized as potentially inappropriate for older adults as they have a higher risk of adverse effects.13 Although numerous studies have established associations between certain drug classes and ACH-like adverse effects, many fail to explicitly attribute these relationships to ACH pathophysiology, leaving a gap in knowledge of the precise mechanism behind observed clinical outcomes.14,15 Despite this, these side effects should not be underestimated, as prolonged exposure may lead to worsening symptoms and potentially contribute to significant mortality. Graves-Morris et al16 conducted a comprehensive systematic review and meta-analysis and assessed the prognostic utility of specific ACB-focused measures on mortality in older individuals and found that ACB was associated with an increased risk of death, with an odds ratio (OR) of about 1.4, in those aged 65 years or older. This increased mortality rate emphasizes the importance of early identification and intervention to ultimately reduce these adverse outcomes in vulnerable populations. Due to the severity of long-term exposure to ACB, numerous complications can arise, and substantial research has been conducted to investigate these outcomes.
A multitude of morbidities may result from a prolonged exposure to ACB. The pathophysiology of ACH action has been associated with the onset of psychosis and psychiatric symptoms in the elderly population with and without dementia.17 Cancelli et al18 conducted a study in patient with Alzheimer’s disease and found that those using ACH medications were 18.3% more likely to have psychosis than those using non-ACH medications, with an OR of 2.52. This association remained significant even after accounting for confounding variables, including worsening cognitive function.18 This association is further supported by another study where serum ACH activity was positively correlated with items of paranoid and delusional ideation and diurnal rhythm disturbances.19 Research has linked psychotic symptoms to metabolic and perfusion alterations in the temporal and frontal cortices, potentially resulting from cholinergic deficits, in addition to elevated binding to M2 receptors in these brain regions.20–22
The association between ACB and dementia has been the subject of extensive research, with numerous clinical studies investigating the potential cognitive impacts of long-term exposure. Epidemiological research has demonstrated that the impact of ACH medications on the development of dementia is significant, with a 30% to 50% increased risk among those with ACB. The associated risk with ACH use has been detected up to 20 years before diagnosis.23–26 In addition, research has revealed a wide array of cognitive impairments associated with ACH medication use in patients with psychiatric disorders, highlighting the extensive effects of these drugs on brain function. Studies have demonstrated that patients with schizophrenia and other severe psychiatric disorders are exposed to high ACH loads exhibiting deficits across multiple cognitive domains, including memory, executive function, and global cognition.27–29
ACB has been associated with a variety of morbidities affecting other parts of the body in addition to its cognitive effects. ACH medications, especially some psychotropics, have been shown to increase the risk of falls and fractures in the elderly due to associated confusion and functional impairment.30,31 In addition, ACB has a critical effect on oral health. There is a significant correlation between ACH medication–induced xerostomia and the increased risk of root caries, dysphagia, and oral microbial changes.32–35 This can adversely affect a patient’s oral hygiene and reduce the number of therapeutic options in dental practice if left untreated. There is also growing evidence of drugs with ACH activity having proarrhythmic, pro-ischemic, and inflammatory effects in the cardiovascular scene. A higher ACB was associated with an increased risk of acute cardiovascular events, including myocardial infarction, strokes, arrythmias, conduction disorders, and cardiovascular death.36–38 ACH medications may contribute to a higher risk of urinary tract infections. Tsubouchi et al15 conducted a meta-analysis that showed that antimuscarinic agents significantly increased incidences of both urinary tract infection and lower urinary tract symptoms and dysfunction. Although there are limited studies regarding this relationship, there is substantial research on the association between urinary retention and urinary tract infections. As urinary retention is a well-documented ACH-related adverse effect, it can be inferred that this class of medications could lead to a higher incidence.39–41
The risk of mortality is generally known to increase with higher ACB, but the strength of this association varies between clinical studies due to the different approaches of quantifying ACB. Graves-Morris et al42 conducted a comprehensive systematic review and meta-analysis and sought to clarify if any particular ACB scale was the most optimal for risk prediction. It found a large evidence base for the Anticholinergic Cognitive Burden Scale (ACB Scale) but was not able to compare superiority as there is a lack of studies comparing different scales.1,42,44,45 Although there is an abundance of tools to measure the extent of ACB, it is still unclear what tool performs best. The ACB Scale is currently the most widely used and accepted scale. It was developed by a panel of experts and has been meticulously validated and studied in a plethora of clinical trials.45 However, although it may account for real-world patient variability and is adaptable, it still has its limitations, as it may be inconsistent, possibly biased, and lacks robust validation in various clinical outcomes. A scale developed by Xu et al,46 the ACH toxicity score (ATS), may provide more objective measures since it relies on muscarinic receptor affinities to scale burden. By using a program that compares known drug affinities and structures, it creates a continuous scale and assigns scores based on affinities to M1, M2, M3, and M4 muscarinic receptors.46 This tool may provide an alternative insight on the correlation of various outcomes with certain ACH scores.
With limited data on the level of ACB in patients with psychiatric disorders, this study aims to address if there is a need for clinical consideration in a vulnerable population. In addition, we hypothesize that due to seasonal prescribing, there would be an increase in the number of patients using anticholinergics, such as allergy medications and antihistamines, and thus there would be an increase in the amount of ACB in the population. By utilizing the ATS, the study looks to measure and compare the degree of ACB across two different time periods. This approach will determine if there is a difference in prescribing patterns of ACH medications throughout the year and whether a significant percentage of patients are exposed to high ACB, which may indicate the need for targeted interventions to mitigate adverse effects.
Methods
This study was a retrospective analysis that collected medication regimens and demographic data from the electronic health records of inpatients at a state psychiatric center. ACB was assessed through the utilization of the ATS from data collected at two distinct time points. The initial time frame data, which included a list of each patient’s medications, were taken from February 1, 2022 (winter), and the second time frame was selected from June 1, 2023 (spring). Only medications that were confirmed to have been administered on these dates, including as-needed (PRN) medications, were included in the analysis. Long-acting injectables (LAIs) that were administered and known to be metabolically active during these specified days were also included in the datasets. A power analysis determined the ideal sample size.
The inclusion criteria included all patients who were institutionalized at the state psychiatric center on the dates of interest and had active scheduled medications. Patients were excluded if they were designated under the state-defined status of criminal procedure law (CPL).
Data analysis. Patient medication lists were analyzed, and ACB scores were calculated based on affinity scores from the ATS with scores divided into M1, M2, and M3 and M4 and M5 affinities prior to being summed up into a total burden score. The data analysis consisted of an analysis of variance (ANOVA) with a probability level set at 0.01.
Results
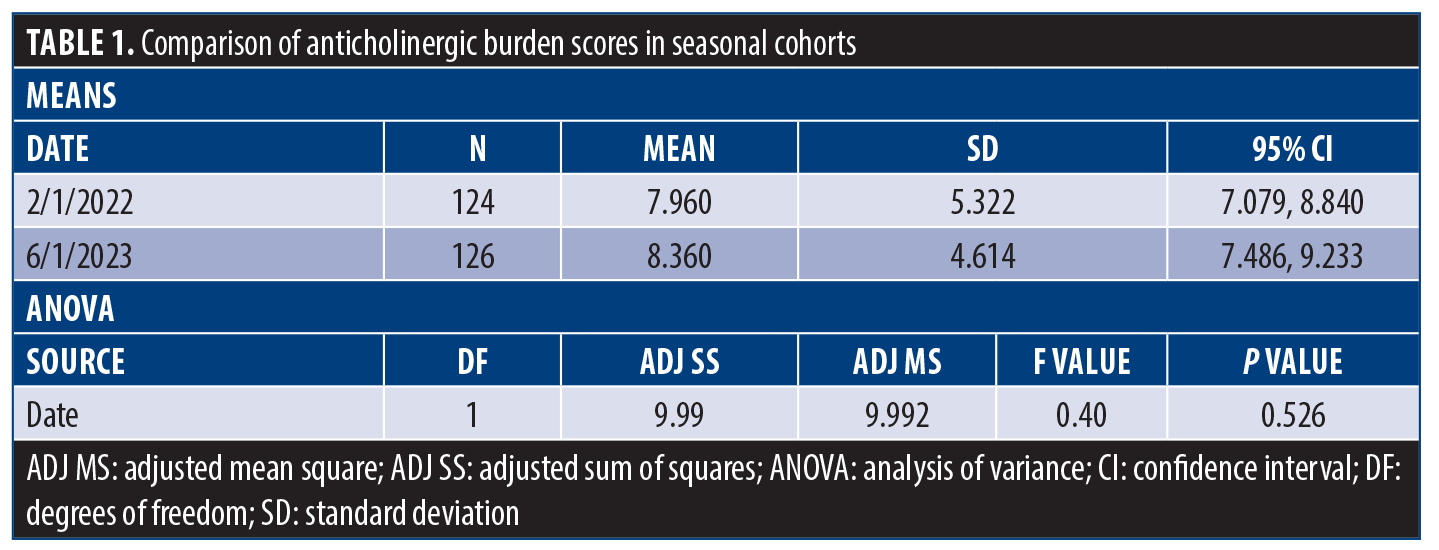
A total of 124 patients were included from the winter of 2022 and 126 patients from the spring of 2023. Out of the total number of patients in the spring and winter, 61 and 49 were on LAIs with duration of action overlapping the dates of interest, respectively. Comparison of ACB scores in seasonal cohorts did not vary significantly between February 1, 2022 (n=124), with a mean ACB score of 7.96, and June 1, 2023 (n=126), with a mean ACB score of 8.36 (Table 1). Patients ranged from 19 to 90 years of age, with most patients being in their late 50s and early 60s. There were 22 and 29 geriatric patients who were older than 65 years of age in the winter and spring cohorts, respectively.
In the winter cohort, 98.4% (122/124) of patients were on antipsychotics, 16.9% (21/124) were on antidepressants, 7.3% (9/124) were on antisecretory agents, 11.3% (14/124) were on antihistamines, and 24.2% (30/124) were on antispasmodics. In the spring cohort, 99.2% (125/126) of patients were on antipsychotics, 27.8% (35/126) were on antidepressants, 5.56% (7/126) were on antisecretory agents, 18.3% (23/126) were on antihistamines, and 28.6% (36/126) were on antispasmodics.
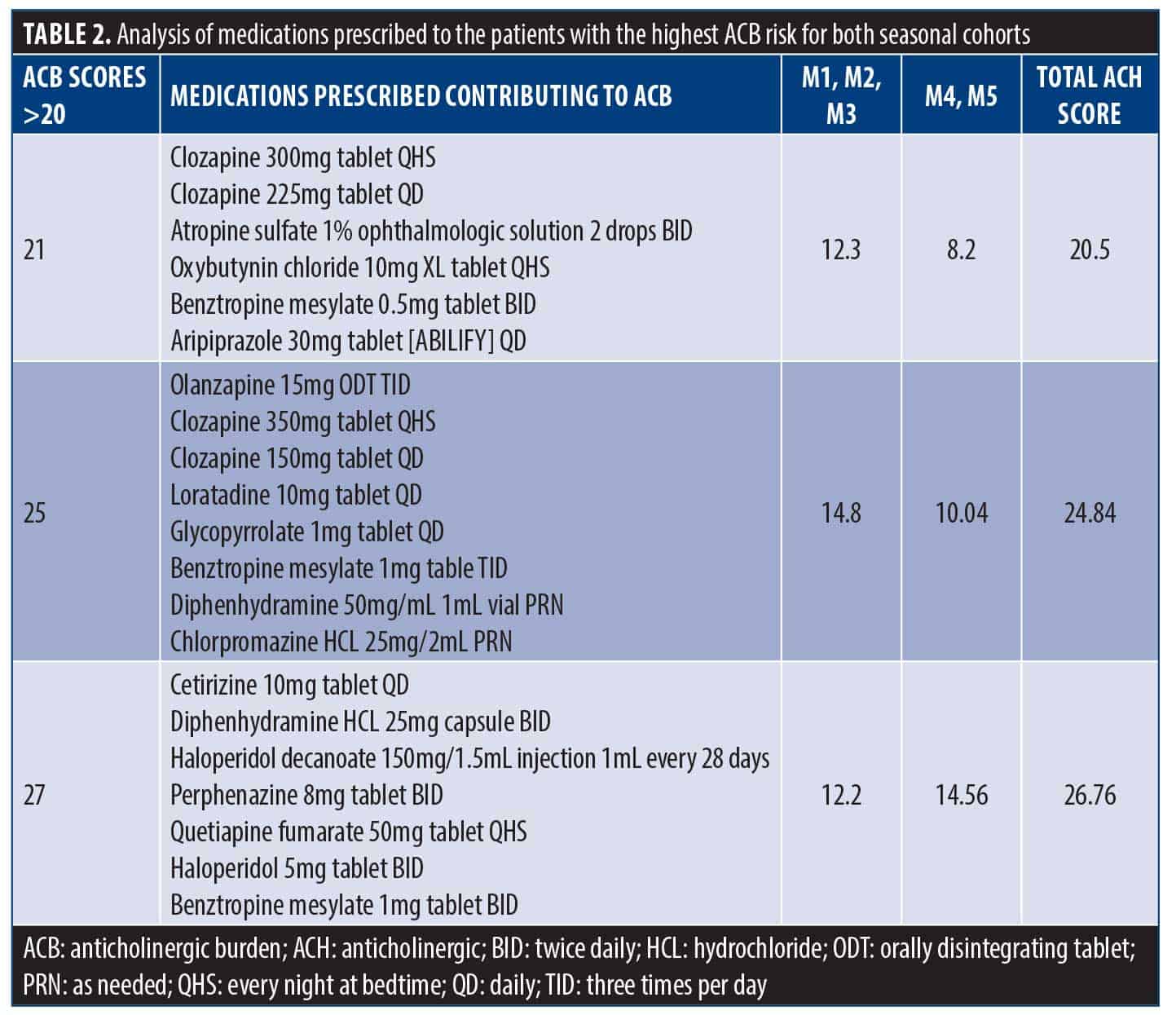
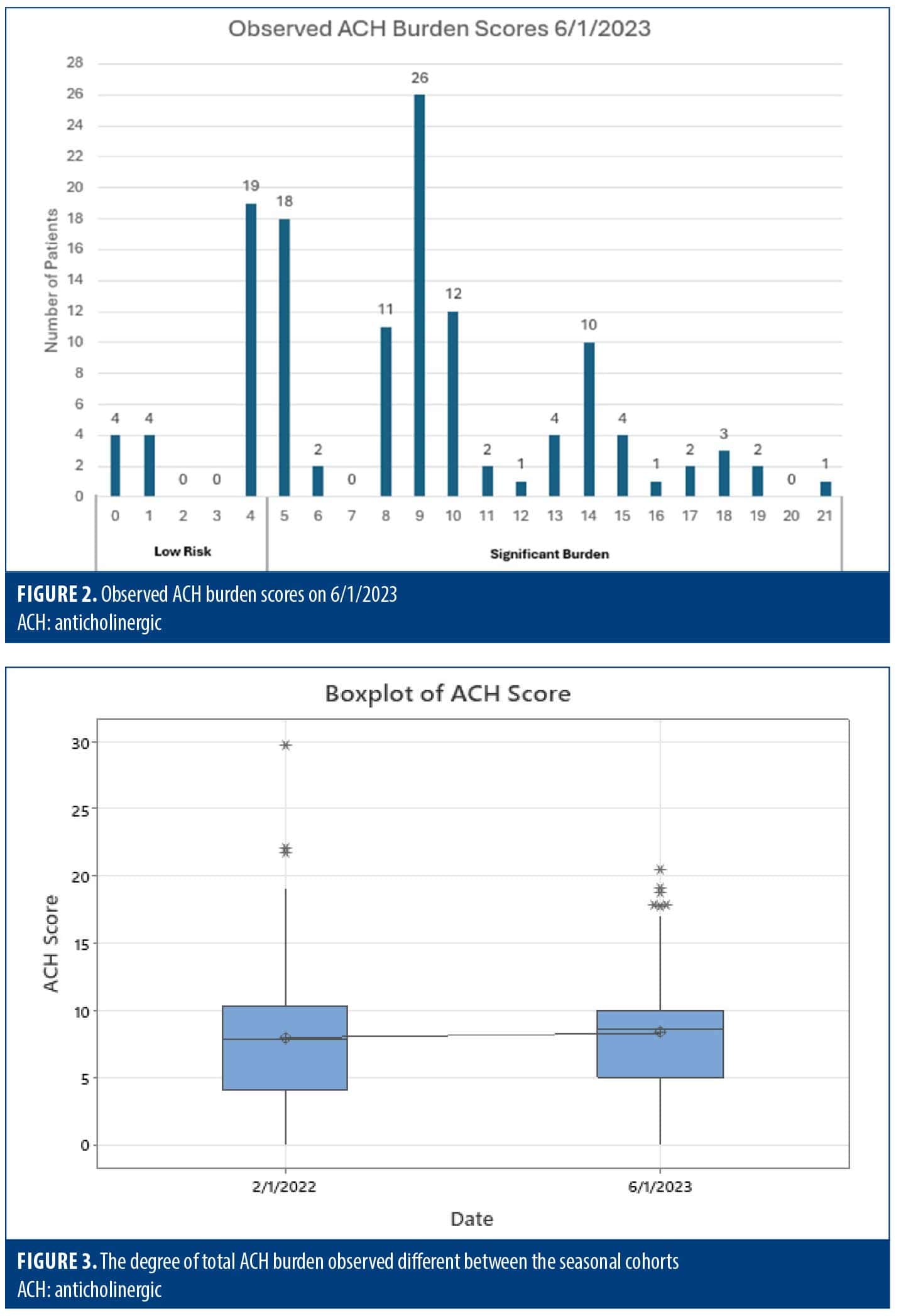
The degree of total ACB was not statistically significantly different between the seasonal cohorts (P=0.526) and remained consistent with an average total ATS score of 8 (Figures 1–3). ANOVA showed no significant difference in the degree of burden between the two groups, and therefore, it appears there was no difference in prescribing trends over time. However, it was found that the majority (70–80%) of inpatients were prescribed multiple ACH medications, with an average ATS of 8 across both cohorts, denoting significant ACB, and thus, these patients were at a higher risk of adverse side effects (Table 2).
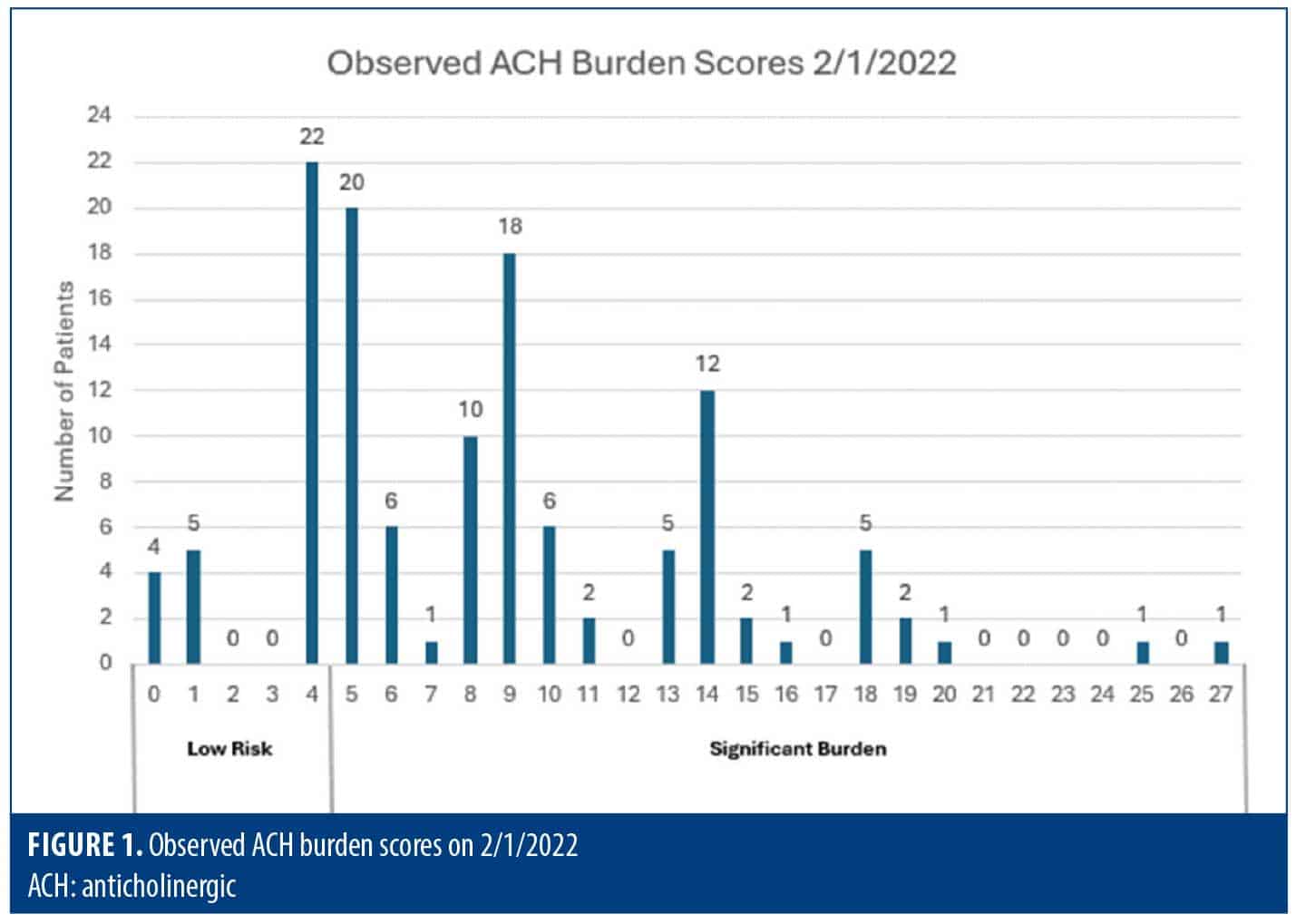
The continuous values of the ATS can be translated into ordinal values for a qualitative comparison with the ACB Scale, according to Xu et al.46 As a result, it can be determined that an ATS of 5 or higher is assumed to be clinically significant. Ninety-three patients (75%) in the winter cohort had clinically significant ACB compared to 99 patients (78.6%) in the spring cohort.
Discussion
This retrospective study extracted drug profiles from inpatients with psychiatric disorders from two distinct seasonal cohorts and evaluated the ACB of administered medications. It was hypothesized that there would be in increase in ACB due to an increase in the prescribing of allergy medications and antihistamines as the seasons change.
The data emphasizes that inpatients with psychiatric disorders are a particularly vulnerable population due to the frequent prescribing of antipsychotics in addition to other ACH medications. This cumulative burden leads to unwanted adverse effects and may negatively affect cognitive functions, which may hinder recovery and overall functionality. Although ACB is prevalent and impacts geriatric patients more severely, studies such as Cebron Lipovac et al7 have shown that up to 25% of adults and 20% of children are exposed to significant burden and that ACB is relevant across all ages. Given sufficient ACB due to prescribing trends, psychiatric inpatients, regardless of age, are at a higher risk of ACH adverse outcomes.
Limitations. This single-center study was limited to the inpatient population of a state psychiatric facility where outpatient tenure could not be maintained. The study was further limited due to the exclusion of patients designated under CPL. Demographics were similar in both groups, with most of the patients identifying as White or African American. As such, the data may not represent and be generalizable to people in the outpatient sector receiving psychiatric medications.
This study utilizes the ATS developed by Xu et al,46 which offers a more objective and nuanced approach to measuring ACB. Although extensively validated and widely used, the ACB Scale is limited by reliance on expert consensus, and thus may introduce bias and have a potential for misclassifications. It may not be an optimal tool to precisely quantify burden or account for individual variability in drug metabolism and sensitivity due to its categorial nature. The ATS is also not without its limitations. Although it boasts a mechanistic approach and a stronger correlation with ACH adverse events, it solely relies on muscarinic binding affinities and lacks integration of other pharmacological properties, such as absorption, distribution, metabolism, and excretion data.
Newer scales, such as the machine learning model from Phutietsile et al,45 claim higher predictive performance by incorporating factors such as bioavailability, antimuscarinic activity, blood-brain barrier penetration, chemical structure, and P-glycoprotein substrate profile. Theoretically, newer drugs would be able to be quickly added to this scale through the input of pharmacological data compared to an evaluation by an expert panel. However, scales such as the ATS and machine learning model still lack real-world applications, and future comparative studies are needed for clinical validation.
Conclusion
Our findings show that there were no differences in prescribing trends of ACH medications among inpatients with psychiatric disorders between two different seasons. However, the majority of this population was found to have significant ACB, which emphasizes the risk of adverse outcomes in this population. The evidence linking ACB to cognitive impairment, dementia risk, and poor outcomes in patients with psychiatric disorders underscores the urgent need for further research into its long-term effects and safer prescribing practices. Given the profound physiological and cognitive impact of ACH medications, addressing this issue is critical to improving recovery, quality of life, and future health outcomes for this vulnerable population.
References
- Soiza RL, Boustani MA, Campbell NL, Mangoni AA. Editorial: deprescribing and minimizing use of anticholinergic medications. Front Pharmacol. 2021;12:820051.
- Lu WH, Wen YW, Chen LK, Hsiao FY. Effect of polypharmacy, potentially inappropriate medications and anticholinergic burden on clinical outcomes: a retrospective cohort study. CMAJ. 2015;187(4):E130E137.
- Keller MS, Qureshi N, Mays AM, et al. Cumulative update of a systematic overview evaluating interventions addressing polypharmacy. JAMA Netw Open. 2024;7(1):e2350963.
- Bidarolli M, Das B, Rawat VS, et al. Polypharmacy and anticholinergic burden scales in older adults: a cross-sectional study among psychiatric outpatients in a tertiary care hospital. BMC Geriatr. 2025;25(1):43.
- Sumukadas D, McMurdo ME, Mangoni AA, Guthrie B. Temporal trends in anticholinergic medication prescription in older people: repeated cross-sectional analysis of population prescribing data. Age Ageing. 2014;43(4):
515–521. - González Peña OI, López Zavala MÁ, Cabral Ruelas H. Pharmaceuticals market, consumption trends and disease incidence are not driving the pharmaceutical research on water and wastewater. Int J Environ Res Public Health. 2021;18(5):2532.
- Cebron Lipovec N, Jazbar J, Kos M. Anticholinergic burden in children, adults and older adults in Slovenia: a nationwide database study. Sci Rep. 2020;10(1):9337.
- Hilmer SN, Gnjidic D. The anticholinergic burden: from research to practice. Aust Prescr. 2022;45(4):118120.
- Peralta V, de Jalón EG, Moreno-Izco L, et al. The effect of anticholinergic burden of psychiatric medications on major outcome domains of psychotic disorders: a 21-year prospective cohort study. Schizophr Res. 2024;264:
386–393. - Heald A, Livingston M, Yung A, De Hert MA. Prescribing in schizophrenia and psychosis: Increasing polypharmacy over time. Hum Psychopharmacol. 2017;32(2):10.1002/hup.2579.
- Costa J, Gonzalez JM, Guzman M. 5PSQ-182 Anticholinergic burden in patients admitted to a psychiatric hospital. Eur J Hosp Pharm. 2021;28(Suppl 1):A145.
- Chahine B, Al Souheil F, Yaghi G. Anticholinergic burden in older adults with psychiatric illnesses: a crosssectional study. Arch Psychiatr Nurs. 2023;44:26–34.
- 2023 American Geriatrics Society Beers Criteria® Update Expert Panel. American Geriatrics Society 2023 updated AGS Beers Criteria® for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2023;71(7):2052–2081.
- O’Donnell C, Demler TL, Trigoboff E. Selective serotonin reuptake inhibitors (SSRIs) and their effect on patient aggression in adult patients in a state psychiatric facility: a retrospective analysis. Innov Clin Neurosci. 2022;19(1–3):
33–38. - Tsubouchi K, Arima H, Abe M, et al. Effect of pharmacotherapy for overactive bladder on the incidence of and factors related to urinary tract infection: a systematic review and meta-analysis. J Urol. 2023;209(4):665–674.
- Graves-Morris K, Stewart C, Soiza RL, et al. The prognostic value of anticholinergic burden measures in relation to mortality in older individuals: a systematic review and meta-analysis. Front Pharmacol. 2020;11:570.
- López-Álvarez J, Sevilla-Llewellyn-Jones J, Agüera-Ortiz L. Anticholinergic drugs in geriatric psychopharmacology. Front Neurosci. 2019;13:1309.
- Cancelli I, Valentinis L, Merlino G, et al. Drugs with anticholinergic properties as a risk factor for psychosis in patients affected by Alzheimer’s disease. Clin Pharmacol Ther. 2008;84(1):63–68.
- Hori K, Konishi K, Watanabe K, et al. Influence of anticholinergic activity in serum on clinical symptoms of Alzheimer’s disease. Neuropsychobiology. 2011;63(3):147–153.
- Lemstra AW, Eikelenboom P, van Gool WA. The cholinergic deficiency syndrome and its therapeutic implications. Gerontology. 2003;49(1):55–60.
- Lai MK, Lai OF, Keene J, et al. Psychosis of Alzheimer’s disease is associated with elevated muscarinic M2 binding in the cortex. Neurology. 2001;57(5):805–811.
- Cummings JL, Back C. The cholinergic hypothesis of neuropsychiatric symptoms in Alzheimer’s disease. Am J Geriatr Psychiatry. 1998;6(2 Suppl 1):S64–S78.
- Coupland CAC, Hill T, Dening T, et al. Anticholinergic drug exposure and the risk of dementia: a nested case-control study. JAMA Intern Med. 2019;179(8):1084–1093.
- Zheng YB, Shi L, Zhu XM, et al. Anticholinergic drugs and the risk of dementia: a systematic review and meta-analysis. Neurosci Biobehav Rev. 2021;127:296–306.
- Chatterjee S, Talwar A, Aparasu RR. Anticholinergic medications and risk of dementia in older adults: where are we now? Expert Opin Drug Saf. 2020;19(10):1251–1267.
- Dmochowski RR, Thai S, Iglay K, et al. Increased risk of incident dementia following use of anticholinergic agents: a systematic literature review and meta-analysis. Neurourol Urodyn. 2021;40(1):28–37.
- Opler MGA, Claxton A, McGrory J, et al. Functioning and cognition in patients with schizophrenia after initiating treatment with aripiprazole lauroxil: secondary outcomes and post hoc analysis. Innov Clin Neurosci. 2024;21(1–3):43–51.
- Lindenmayer JP, Insel BJ, Khan A, et al. Effects of clozapine on neurocognitive functions in schizophrenia: a naturalistic comparison to non-clozapine antipsychotics. Innov Clin Neurosci. 2021;18(10–12):40–46.
- Harvey PD, Bowie CR. Cognitive remediation in severe mental illness. Innov Clin Neurosci. 2012;9(4):27–30.
- Ruxton K, Woodman RJ, Mangoni AA. Drugs with anticholinergic effects and cognitive impairment, falls and all-cause mortality in older adults: a systematic review and meta-analysis. Br J Clin Pharmacol. 2015;80(2):
209–220. Erratum in: Br J Clin Pharmacol. 2015;80(4):921–926. - Hilmer SN, Gnjidic D. The anticholinergic burden: from research to practice. Aust Prescr. 2022;45(4):118120.
- Kakkar M, Caetano de Souza Valentim E, Barmak AB, Arany S. Potential association of anticholinergic medication intake and caries experience in young adults with xerostomia. J Dent Sci. 2023;18(4):16931698.
- Cockburn N, Pradhan A, Taing MW, et al. Oral health impacts of medications used to treat mental illness. J Affect Disord. 2017;223:
184–193. - Prado-Mel E, Ciudad-Gutiérrez P, Rodríguez-Ramallo H, et al. Association between anticholinergic activity and xerostomia and/ or xerophthalmia in the elderly: systematic review. BMC Pharmacol Toxicol. 2022;23(1):94.
- Barbe AG. Medication-induced xerostomia and hyposalivation in the elderly: culprits, complications, and management. Drugs Aging. 2018;35(10):877–885.
- Sanghavi R, Pana TA, Mamayusupova H, et al. Higher anticholinergic burden from medications is associated with significant increase in markers of inflammation in the EPIC-Norfolk prospective population-based cohort study. Br J Clin Pharmacol. 2022;88(7):3297–3306.
- Huang WC, Yang AS, Tsai DH, et al. Association between recently raised anticholinergic burden and risk of acute cardiovascular events: nationwide case-case-time-control study. BMJ. 2023;382:e076045.
- Myint PK, Fox C, Kwok CS, et al. Total anticholinergic burden and risk of mortality and cardiovascular disease over 10 years in 21,636 middle-aged and older men and women of EPIC-Norfolk prospective population study. Age Ageing. 2015;44(2):219–225.
- Afonso AS, Verhamme KM, Stricker BH, et al. Inhaled anticholinergic drugs and risk of acute urinary retention. BJU Int. 2011;107(8):
1265–1272. - Stoniute A, Madhuvrata P, Still M, et al. Oral anticholinergic drugs versus placebo or no treatment for managing overactive bladder syndrome in adults. Cochrane Database Syst Rev. 2023;5(5):CD003781.
- Stoffel JT, Peterson AC, Sandhu JS, et al. AUA white paper on nonneurogenic chronic urinary retention: consensus definition, treatment algorithm, and outcome end points. J Urol. 2017;198(1):153–160.
- Graves-Morris K, Stewart C, Soiza RL, et al. The prognostic value of anticholinergic burden measures in relation to mortality in older individuals: a systematic review and meta-analysis. Front Pharmacol. 2020;11:570.
- Hanlon P, Quinn TJ, Gallacher KI, et al. Assessing risks of polypharmacy involving medications with anticholinergic properties. Ann Fam Med. 2020;18(2):148–155.
- Salahudeen MS, Duffull SB, Nishtala PS. Anticholinergic burden quantified by anticholinergic risk scales and adverse outcomes in older people: a systematic review. BMC Geriatr. 2015;15:31.
- Phutietsile GO, Fotaki N, Nishtala PS. Assessing the anticholinergic cognitive burden classification of putative anticholinergic drugs using drug properties. Br J Clin Pharmacol. 2024;90(9):2236–2255.
- Xu D, Anderson HD, Tao A, et al. Assessing and predicting drug-induced anticholinergic risks: an integrated computational approach. Ther Adv Drug Saf. 2017;8(11):361–370.
