 by Michael M. Messer, MD, and Irina V. Haller, PhD, MS
by Michael M. Messer, MD, and Irina V. Haller, PhD, MS
Dr. Messer is with Behavioral Health Department, Essentia Health, Duluth, MN, and Dr. Haller is with Essentia Institute of Rural Health, Essentia Health, Duluth, MN, USA.
Innov Clin Neurosci. 2017;14(1–2):56–59.
Funding: This research had no grant funding from any funding agency in the public, commercial, or non-profit sectors.
Financial disclosures: The authors have no conflicts of interest relevant to the content of this article.
Key words: Multiple sclerosis, major depressive disorder, treatment-resistant depression, ketamine, electroconvulsive therapy
Abstract: Objective—Depression is a common condition among patients with multiple sclerosis and often becomes resistant to oral antidepressants. We report a patient with multiple sclerosis who developed severe treatment-resistant depression and who was successfully treated with intravenous ketamine over the period of two years.
Methods—Ketamine treatment protocol included an initial series of six treatments administered every other day, followed by a maintenance schedule. Ketamine was administered intravenously at 0.5mg/kg of ideal body weight over 40 minutes. Depression symptoms were measured using Beck Depression Index.
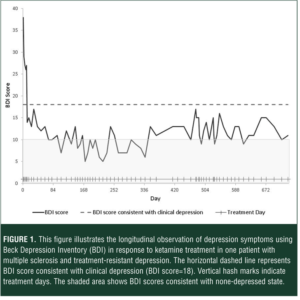
Results—The patient’s Beck Depression Index score prior to initiating ketamine treatment was 38, corresponding to severe depression. Response to treatment, defined as 50-percent reduction in Beck Depression Index score, was observed after five treatments. For this patient, the maintenance schedule ranged from a weekly treatment to one treatment every three weeks. During the two-year observation period, this patient was able to maintain a stable non-depressed mood and had no worsening of her MS symptoms.
Conclusion—Ketamine may be an alternative treatment for resistant depression and may have a special use in patients with multiple sclerosis.
Introduction
Depression is a frequent finding in patients with multiple sclerosis (MS), with the lifetime prevalence rates for major depressive disorder (MDD) ranging from 36 to 54 percent, more than twice of that in the general population.[1] Even with advances in pharmacological options for treating depression, an estimated 33 to 66 percent of patients with MDD in the general population do not respond to the first antidepressant, and a reported 15 to 33 percent of patients do not respond to multiple interventions.[2] For patients with severe or treatment-resistant depression unsuccessfully treated with multiple pharmacological options, electroconvulsive therapy (ECT) is often the only available treatment.
A recent review summarized cases of 21 MS patients with various psychiatric conditions treated with ECT and provided limited evidence for effective and safe use of ECT in these patients.[3–5] The majority of these cases reported improved psychiatric status (19/21) and unchanged neurologic status after ECT (17/21).[3] Negative neurologic effects in these patients included delirium, urinary incontinence, muscle weakness, and seizures.[6,7] Despite the apparent efficacy of ECT in treating psychiatric illness in MS patients, long-term effects, including autonomic instability and neurologic deterioration, following ECT are still unclear.[4,5,7,8]
Ketamine, a N-methyl-D-aspartate (NMDA) receptor antagonist emerged as a potential treatment of mood disorders with rapid and sustained antidepressant effect.[9] At low doses for antidepressant treatment, side effects associated with ketamine include perceptual disturbances, confusion, blood pressure elevation, euphoria, dizziness, and increased libido, but these symptoms usually resolve within 80 minutes after ketamine infusion.[10]
We present a case of a patient with MS and TRD. She was initially considered for ECT after not responding to oral antidepressants, but her depression was successfully treated with intravenous ketamine. Shehad maintenance of the recovery from depression over two years with significant reduction of her depressive symptoms. This observation suggests a potential alternative to ECT in treating TRD in a patient with MS.
Case report
In September 2007, a 45-year-old woman was referred to psychiatry for worsening depressive symptoms. She had been diagnosed with MS in 2004, presenting with initial symptoms of left arm and hand numbness. Her magnetic resonance imaging (MRI) scan revealed 4 to 5 periventricular white matter lesions and an anterior cervical involvement at C5. She had two exacerbations of her symptoms during the following three months, fulfilling the diagnosis criteria of relapsing remitting MS, and was enrolled in the Beyond medication trial (ID: NCT00099502), using betaseron, in January 2005.
The Beck Depression Inventory (BDI) was used for evaluating her level of depression. At screening for the trial ,the patient’s BDI score was 18, corresponding to borderline clinical depression. She worsened to moderate depression (23 on BDI) by May 2005, but continued her participation in the trial until fall of 2006, when she was diagnosed with MDD. Prior to referral to psychiatry, she was treated with paroxetine, but it did not improve her mood.
From the end of her participation in the Beyond trial to her time of presentation, she did not have any relapses in her MS symptoms, and her MRI and neurologic examinations were stable. Her depression, however, continued to worsen. Between 2007 and 2014, she received courses of five different oral antidepressants (desipramine, venlafaxine, mirtazapine, paroxetine, and buproprion). She had augmentation of the antidepressants without benefit. In January 2014, she requested ECT from psychiatry due to apparent TRD. Due to concern for her neurologic condition and potential risk of worsening her MS symptoms, ketamine treatment was discussed and initiated as an alternative to ECT.
Our clinic has more than eight years of experience in treating TRD with ketamine.[11,12] The treatment protocol includes an initial series of six treatments administered every other day, followed by a maintenance schedule starting with a weekly treatment and tapering to one treatment every three weeks.[11] Ketamine is administered intravenously at 0.5mg/kg of ideal body weight over 40 minutes.
The longitudinal observation of depression symptoms (measured by BDI) in response to ketamine treatments in this patient is shown in the Figure 1. She received her first treatment in January 2014, and was followed for two years using our established protocol. Her BDI score before initiation of the treatment was 38, corresponding to severe depression. Response to treatment, defined as 50-percent reduction in BDI scores, was observed after the fifth treatment (Day 10) with a BDI score of 19, corresponding to borderline clinical depression. The calibration of treatment frequency for maintenance schedule began two weeks after ketamine treatment initiation. For this patient ,the maintenance schedule ranged from one weekly treatment to one treatment every three weeks. During the maintenance period, the BDI scores ranged between 5 and 16, corresponding to non-depressed state. The response to treatment was sustained through the observation period with reduction of depressive symptoms compared to scores prior to initiation of ketamine treatment consistently ranging between 63 percent and 95 percent of the baseline BDI score.
The clinical course for this patient followed a similar trajectory as seen with other patients who have TRD. The presence of MS did not make her more or less sensitive to the antidepressant effects of ketamine. Her antidepressant medications remained at a stable dose throughout the course of treatment. She was able to maintain a stable and non-depressed mood with this treatment and to date has had no worsening of her MS symptoms.
Discussion
Depression is common in patients with MS, and some patients may become resistant to oral medications.[1] Generally, TRD may be defined as a failure to respond to at least two different types of oral antidepressant treatments for a period longer than four weeks at the maximum recommended dose. ECT is often used for TRD; however, based on the limited number of published cases there are still questions about its safety in patients with MS, and the long-term effects of ECT in this population are still unclear.[3] Moreover, most current treatments for TRD take several weeks to achieve clinical effect. In comparison, ketamine has a rapid onset of action.10
Glutamatergic system plays a role in neuronal plasticity and cellular resiliency, and a growing body of evidence also suggests that it is also involved in the pathophysiology and treatment of MDD.[13,14] Ketamine, a potent selective NMDA receptor antagonist with rapid antidepressant effect even in small doses, has emerged as a promising agent for treating TRD.[10] Glutamate systems have also been associated with stroke, traumatic brain injuries, Alzheimer’s disease, and amyotrophic lateral sclerosis (ALS).[9] Another NMDA receptor antagonist, riluzole, with less potency than ketamine, has been approved for ALS treatment, but is also used for treatment of TRD.[15] Ketamine has also been safely used to treat severe pain and allodynia in a patient with MS.[16] It was reported to have an anti-inflammatory effect by suppression of NF-kappaB, which is likely involved in brain neurodegenerative inflammatory diseases, such as MS, suggesting that ketamine may be neuroprotective.[17]
Side effects of ketamine treatment for TRD can include transient hypertension during the infusion, anxiety, nausea or dizziness, euphoria or giddiness, pressured speech, and dissociative symptoms. Dissociation appears to be more likely when higher doses are used. Dissociation is expressed as “losing control” or being focused on an idea or thought that might be distressing. We have seen a patient with posttraumatic stress disorder who became very distressed by the repressed thoughts, which surfaced after the ketamine infusion. To avoid dissociative symptoms, we have adopted the practice of using the “ideal” body weight for dosing. This was noted after a patient, twice his ideal body weight, had profound dissociative symptoms with visual distortions.[12] Thus, starting dosing at 0.5mg/kg of ideal body weight and advancing as needed for antidepressant effect helps to mitigate dissociative symptoms.
The access to treatment with ECT must also be taken into consideration when it becomes the primary antidepressant intervention. While ECT is only available in specialized centers, ketamine treatments can be done safely in an outpatient setting, infusion center, or hospital. Compared to ECT, this makes ketamine a more accessible treatment for TRD. Also, initiating treatment using an intravenous route of administration allows observation of the patient’s response and interruption of treatment in case of adverse events. If a patient does well with intravenous treatments, the route of administration could be changed to intramuscular injections if such change is clinically indicated or more convenient to the patient. Parenteral use of ketamine prevents diversion or abuse and keeps the control of the medication with the treating physician.
Conclusion
In the case described here, a patient with MS and TRD was successfully treated with intravenous ketamine for her depression over two years and had significant reduction of depressive symptoms. Thus, ketamine may be an alternative treatment to ECT for resistant depression. In particular, ketamine may have a special use in patients with MS and TRD, as ECT has the potential for side effects in this patient population.
Acknowledgment
Patient consent to publish this report was obtained.
References
- Minden SL, Feinstein A, Kalb RC, et al. Evidence-based guideline: assessment and management of psychiatric disorders in individuals with MS: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2014;82(2):174–181.
- Little A. Treatment-resistant depression. Am Fam Physician. 2009;80(2):167–172.
- Palm U, Ayache SS, Padberg F, Lefaucheur JP. Non-invasive brain stimulation therapy in multiple sclerosis: a review of tDCS, rTMS and ECT results. Brain Stimul. 2014;7(6):849–854.
- Fitzsimons MG, Welch CA, Haspel KL, Gorman JM. The safety and efficacy of ECT and anesthesia in the setting of multiple sclerosis. J Psychiatr Pract. 2007;13(3):195–198.
- Rasmussen KG, Keegan BM. Electroconvulsive therapy in patients with multiple sclerosis. J ECT. 2007;23(3):179–180.
- Mattingly G, Baker K, Zorumski CF, Figiel GS. Multiple sclerosis and ECT: possible value of gadolinium-enhanced magnetic resonance scans for identifying high-risk patients. J Neuropsychiatry Clin Neurosci. 1992;4(2):145–151.
- Urban-Kowalczyk M, Rudecki T, Wroblewski D, et al. Electroconvulsive therapy in patient with psychotic depression and multiple sclerosis. Neurocase. 2014;20(4):452–455.
- Pontikes TK, Dinwiddie SH. Electroconvulsive therapy in a patient with multiple sclerosis and recurrent catatonia. J ECT. 2010;26(4):270–271.
- Serafini G, Howland RH, Rovedi F, et al. The role of ketamine in treatment-resistant depression: a systematic review. Curr Neuropharmacol. 2014;12(5):444–461.
- Zarate CA, Jr., Singh JB, Carlson PJ, et al. A randomized trial of an N-methyl-D-aspartate antagonist in treatment-resistant major depression. Arch Gen Psychiatry. 2006;63(8):856–864.
- Messer M, Haller IV. Maintenance ketamine treatment produces long-term recovery from depression. Primary Psychiatry. 2010;17(4):54–56.
- Messer M, Haller IV, Larson P, et al. The use of a series of ketamine infusions in two patients with treatment-resistant depression. J Neuropsychiatry Clin Neurosci. 2010;22(4):442–444.
- Paul IA, Skolnick P. Glutamate and depression: clinical and preclinical studies. Ann N Y Acad Sci. 2003;1003:250–272.
- Zarate CA, Jr., Du J, Quiroz J, et al. Regulation of cellular plasticity cascades in the pathophysiology and treatment of mood disorders: role of the glutamatergic system. Ann N Y Acad Sci. 2003;1003:273–291.
- Zarate CA, Manji HK. Riluzole in psychiatry: a systematic review of the literature. Expert Opin Drug Metab Toxicol. 2008;4(9):1223–1234.
- Sakai T, Tomiyasu S, Ono T, et al. Multiple sclerosis with severe pain and allodynia alleviated by oral ketamine. Clin J Pain. 2004;20(5):375–376.
- Sakai T, Ichiyama T, Whitten CW, et al. Ketamine suppresses endotoxin-induced NF-kappaB expression. Can J Anaesth. 2000;47(10):1019–1024.