 by Mitali Wadekar, MD; Anil Sharma, MD, MBA; and Gina Battaglia, PhD
by Mitali Wadekar, MD; Anil Sharma, MD, MBA; and Gina Battaglia, PhD
Drs. Wadekar and Sharma are with IRB Company, Inc., Buena Park, California, and Dr. Bittaglia is an independent medical writer/consultant from Westlake Village, California.
Innov Clin Neurosci. 2015;12(3–4):27–31.
This ongoing column explores off-label or emerging treatment options and drug development trends in the field of neuroscience.
Funding: No funding was provided for the preparation of this article.
Financial disclosures: The authors have no conflicts of interest relevant to the content of this article.
Key words: Patient-centered outcomes, decision-making capacity, impaired decision-making capacity, central nervous system disorders, CNS disorders, drug development
Abstract: Background: Patient-centered outcomes research increases patient involvement in health-related decisions with better information of benefits, risks, and options as it pertains to patient health. Patient-centered outcomes research is valuable for improving patient options in general; however, the vulnerability of patients with certain central nervous system conditions and their variable capacity to consent may pose significant challenges. Methods/review: Authors acknowledge the historical issues and address needs regarding better dissemination of knowledge in central nervous system conditions like dementia, depression, and schizophrenia. Authors propose ethical safeguards necessary to carry out sound patient-centered outcomes research in this unique population. Discussion/recommendations: To facilitate ethical patient-centered outcomes research, researchers must 1) assess the individual’s mental capacity to provide consent, 2) determine vulnerability and risk of conducting research in specific subgroups, and 3) consider appointment of legally authorized representatives and/or consent monitors to ensure accurate understanding of research and ongoing direct communication with patients, especially when their future prognosis is unpredictable.
Introduction
Patient-centered outcomes research (PCOR), first defined by the Patient-Centered Outcomes Research Institute in March 2011, increases patient involvement in their healthcare by allowing them to evaluate the outcomes of healthcare practices regarding individual preferences, values, and experiences in choosing the optimal treatment plan.[1] PCOR not only improves healthcare quality by providing clear and impartial evidence on the effectiveness, benefits, and risks, but also minimizes harms of treatment options available to patients and physicians because of the considerations of individual patient needs. PCOR increases patient decision-making capability, eliminates health disparities, and lowers healthcare costs.[2]
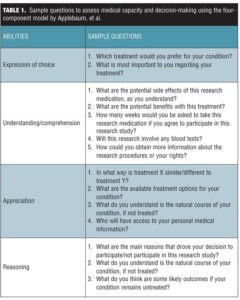
However, certain central nervous system (CNS) conditions, such as dementia, depression, and schizophrenia, may impair an individual’s decision-making capacity (DMC) and thus his or her ability to choose the best treatment. A frequently used assessment model proposed by Applebaum and Grisso[3] addresses four abilities for determining whether an individual possesses sufficient DMC (Table 1):
1. Expression of choice
2. Understanding/comprehension
3. Appreciation
4. Reasoning.
While insufficiency in one or more of these abilities may render a patient less able to independently exercise effective treatment decisions, judgment of DMC may vary among physicians and caregivers.[4,5] DMC impairments also vary widely within CNS disorders such as schizophrenia,[6] and some conditions such as depression may not impair understanding[7] but may alter coherence of personal preference (e.g., an individual’s treatment preference may be different during depressive periods than during nondepressive periods[8]).
Federal agencies and institutional review board (IRB) committees recognize that because CNS patients are a vulnerable population, they require special protection in research studies[9–13] due to their impaired capacity to provide consent[14] and their susceptibility to coercion by study investigators.[9,15] However, these regulations may prevent the approval of valuable research that could improve treatment options in these individuals and reduce the autonomy of these individuals in the decision-making process.[16] The purpose of this article is to discuss the importance of PCOR research in individuals with certain CNS conditions, such as dementia, schizophrenia, and depression, and how IRB committees and researchers should implement safeguards for ethical research in these unique populations.
Methods/Review
Probably the most renowned case of unethical research in individuals with reduced DMC occurred during World War II when Nazi doctors used mentally impaired individuals, as well as prisoners in concentration camps, to conduct medical experiments.[17] The resulting Doctors’ Trial[18] found 16 defendants guilty and led to the implementation of the Nuremberg Code, which requires that participants have legal capacity to give voluntary consent, that subjects can decline participation at any point throughout the experiment, and that the experiments are beneficial to society.[19]
Many unethical research experiments have been undertaken in individuals with CNS conditions both before and after implementation of the Nuremberg Code, including the deliberate infection of Michigan mental institution patients with influenza[20] and of insane asylum patients, prison inmates, and soldiers with syphilis in Guatemala.[21] Mentally handicapped children were also subjected to radiation experiments at the Sonoma State Hospital in northern California[22] and at the Walter E. Fernald State School without parental knowledge or consent.[23] In 1978, the Department of Health, Education, and Welfare implemented federal regulations to provide special protections and restrictions for institutionalized individuals with psychiatric disorders.[9] Although these regulations were not formally enacted due to their stigmatizing nature and their potential to undercut research, further federal regulations were subsequently implemented to protect these individuals in the research setting.[10–12]
Although the Department of Health and Human Services currently does not have specific regulations for research in patients with psychiatric disorders, most current IRB committees have different standards for assessing risk and direct benefit in these individuals than for studies in adults without such CNS disorders.[24] However, these rigorous standards may also lead to overestimation of risk by IRB committees.[25] Many IRB members lack experience with these ethically challenging protocols,[26] and research in CNS populations is often stigmatized.[27] All of these factors may impede the approval of potentially valuable research in this population. Therefore, careful risk/benefit analyses considering the population of interest will promote a greater scope of research in CNS populations and provide an ethical basis for IRB members to review future studies in a more expedited manner.
Discussion and Recommendations
To facilitate ethical PCOR, researchers must assess each individual’s capacity to provide consent. Researchers should be aware that many patients, such as those with Alzheimer’s disease[28] and schizophrenia,[29] may be susceptible to errors in comprehension and communication of their treatment decisions, and that comprehension and communication abilities vary widely even within a given condition. Therefore, they should target their communication to the population of interest to minimize characteristic difficulties.
CNS conditions often tend to have characteristic reductions in abilities. Individuals with schizophrenia score lower on measures of understanding than do patients with depression[30,31] and may be particularly susceptible to coercion.[32] Additionally, many patients with depression lack appreciative ability[33–36] and have a degree of separation between their emotional and cognitive states, thus exhibiting different treatment preferences during active depression and their nondepressive state.[8] Therefore, modifying the consent process based on the condition, such as assessing treatment preferences during both nondepressive and depressive periods, helps protect these individuals against potentially harmful treatment decisions while preserving their autonomy.[8]
Researchers and IRB committees must also assess vulnerability and relative risk on a continuum, considering factors such as the patient setting (e.g., individuals in institutional or correctional settings may be coerced into certain treatments because of their restricted autonomy[37]) and the patient’s DMC, which may be reduced considerably during the active stage of the disorder. Socioeconomic factors may also increase vulnerability and coercion. For example, individuals with active substance abuse problems who need money to purchase drugs may be coerced into a research study in which they can obtain the drugs for free.[38] Thus, researchers and IRB committees should assess vulnerability and risk based on participant condition (including whether they are in the active or stable phase of the disorder), their residential setting, and potential coercions. IRB committee members who are familiar with research in individuals with CNS conditions may be particularly valuable in determining risk in a given population and may be able to expedite minimal-risk research in less vulnerable populations. Consent monitors may be employed by the IRB in studies involving greater than minimal risk to ensure that vulnerable populations are not involuntarily recruited into potentially harmful research.[16]
Finally, individuals with CNS conditions such as dementia that impair DMC may require a legally authorized representative (LAR) to effectively communicate the treatment options and act in the best interests of the participant. Involving the individual in his or her treatments through shared decision making in which the LAR offers support and opinions when the individual questions his or her own judgment may be optimal for involving the individual in his or her treatment decision in cases of moderate dementia.[39] Mental competence may also depend on the complexity of the pending decisions,[40] how a person is understood or treated,[41] or even how an individual is feeling on the particular day.[42] To increase DMC and improve awareness on the impact of the individual’s decisions in PCOR, the LAR should be able to identify and facilitate the retained abilities of the individual and understand the individual’s personal values.
Conclusion
In summary, PCOR is valuable for improving treatment options in patients with CNS conditions and increasing their involvement in their health-related decisions. The potential vulnerability and impaired DMC of individuals with certain CNS conditions, such as dementia, depression, and schizophrenia, have limited the scope of research in this population. IRB members and researchers should carefully assess the consent-providing ability of patients and vulnerability in specific subgroups and employ LARs and/or consent monitors as needed to ensure that the personal values and interests of patients are maintained throughout the study.
References
1. Patient-Centered Outcomes Research Institute. Patient-Centered Outcomes Research. http://www.pcori.org/research-we-support/pcor/. Accessed May 28, 2014.
2. Brigham and Women’s Hospital. Patient-Centered Comparative Effectiveness Research Center. http://www.brighamandwomens.org/research/centers/pcerc/default.aspx. Accessed May 28, 2014
3. Appelbaum PS, Grisso T. The MacArthur Treatment Competence Study. I: mental illness and competence to consent to treatment. Law Hum Behav. 1995;19:105–126.
4. Kim SY, Appelbaum PS, Swan J, et al. Determining when impairment constitutes incapacity for informed consent in schizophrenia research. Br J Psychiatry. 2007;191:38–43.
5. Stocking CB, Hougham GW, Danner DD, et al. Variable judgments of decisional capacity in cognitively impaired research subjects. J Am Geriatr Soc. 2008;56:1893–1897.
6. Jeste DV, Depp CA, Palmer BW. Magnitude of impairment in decisional capacity in people with schizophrenia compared to normal subjects: an overview. Schizophr Bull. 2006;32:121–128.
7. Appelbaum PS, Grisso T, Frank E, et al. Competence of depressed patients for consent to research. Am J Psychiatry. 1999;156:1380–1384.
8. Rudnick A. Depression and competence to refuse psychiatric treatment. J Med Ethics. 2002;28:151–155.
9. The National Commission for the Protection of Human Subjects of Biomedical and Behavioral Research. Research involving those institutionalized as mentally infirm. United States Department of Health, Education and Welfare Publication No (OS) 78-0006;1978. http://videocast.nih.gov/pdf/ohrp_research_mentally_infirm.pdf. Accessed April 1, 2015.
10. National Bioethics Advisory Commission. Research Involving Persons with Psychiatric Disorders that May Affect Decision Making Capacity. Washington, DC: National Bioethics Advisory Commission; 1999. Available at: https://bioethicsarchive.georgetown.edu/nbac/capacity/volumeii.pdf. Accessed May 28, 2014.
11. National Institutes of Health. Office of Extramural Research. Research involving individuals with questionable capacity to consent: points to consider. November 2009. http://grants.nih.gov/grants/policy/questionablecapacity.htm. Accessed May 28, 2014.
12. United States Department of Health and Human Services. Institutional Review Board Guidebook. Chapter VI: Special classes of subjects. 1993. http://www.hhs.gov/ohrp/archive/irb/irb_chapter6.htm. Accessed May 28, 2014.
13. US Department of Health and Human Services. Office for Human Research Protections. Request for information and comments on research that involves adult individuals with impaired decision-making capacity. Federal Register. 2007;72:50966–50970.
14. Levine C, Faden R, Grady C, et al. The limitations of “vulnerability” as a protection for human research participants. Am J Bioeth. 2004;3:44–49.
15. Silvers A. Historical vulnerability and special scrutiny: precautions against discrimination in medical research. Am J Bioeth. 2004;3:56–57.
16. Yanos PT, Stanley BS, Greene CS. Research risk for persons with psychiatric disorders: a decisional framework to meet the ethical challenge. Psychiatr Serv. 2009;60:374–383.
17. Shuster E. Fifty years later: the significance of the Nuremberg Code. N Engl J Med. 1997;337:1436–1440.
18. International Military Tribunal. Trials of war criminals before the Nuremberg Military Tribunals under Control Council law no. 10. Washington, DC: Government Printing Office, 1950.
19. Annas GJ, Grodin MA (eds). The Nazi Doctors and the Nuremberg Code: Human Rights in Human Experimentation. New York: Oxford University Press; 1992.
20. Meiklejohn GN. Commission on influenza. In: Woodward TE (ed). The Armed Forces Epidemiological Board: Histories of the Commissions. Washington, DC: Borden Institute. Office of the Surgeon General. United States Department of the Army; 1994.
21. United States Department of Health and Human Services. Fact sheet on the 1946-1948 U.S. Public Health Service Sexually Transmitted Diseases (STD) Inoculation Study. http://www.hhs.gov/1946inoculationstudy/factsheet.html. Accessed May 28, 2014.
22. Alliance for Human Research Protection. Children were the raw material of medical research. CBS 60 Minutes. Monday, February 28, 2005. http://www.ahrp.org/infomail/05/02/28.php. Accessed May 28, 2014.
23. Blau SE. Institute charged in Fernald radiation experiment lawsuit. The Tech: Online Edition. 1995;115. http://tech.mit.edu/V115/N49/radiation.49n.html. Accessed May 28, 2014.
24. American Psychiatric Association’s Task Force on Research Ethics. Ethical principles and practices for research involving human participants with mental illness. Psychiatric Services. 2006;57:552–557.
25. Michels R. Are research ethics bad for our mental health? N Engl J Med. 1999;340:1427–1430.
26. Sirotin N, Wolf LE, Pollack LM, et al. IRBs and ethically challenging protocols: views of IRB chairs about useful resources. IRB. 2010;32:10–19.
27. Muroff JR, Hoerauf SL, Kim SY. Is psychiatric research stigmatized? An experimental survey of the public. Schizophr Bull. 2006;32:129–136.
28. Marson DC, Annis SM, McInturff B, et al. Error behaviors associated with loss of competency in Alzheimer’s disease. Neurology. 1999;53:1983–1992.
29. Kaup AR, Dunn LB, Saks ER, et al. Decisional capacity and consent for schizophrenia research. IRB. 2011;33:1–9.
30. Stiles PG, Poythress NG, Hall A, et al. Improving understanding of research consent disclosures among persons with mental illness. Psychiatr Serv. 2001;52:780–785.
31. Cohen BJ, McGarvey EL, Pinkerton RC, Kryzhanivska L. Willingness and competence of depressed and schizophrenic inpatients to consent to research. J Am Acad Psychiatry Law. 2004;32:134–143.
32. Anderson KK, Mukherjee SD. The need for additional safeguards in the informed consent process in schizophrenia research. J Med Ethics. 2007;33:647–650.
33. Leeman CP. Depression and the right to die. Gen Hosp Psychiatry. 1999;21:112–115.
34. Halpern J. When concretized emotion-belief complexes derail decision making capacity. Bioethics. 2012;26:108–116.
35. Young EW, Corby JC, Johnson R. Does depression invalidate competence? Consultants’ ethical, psychiatric, and legal considerations. Camb Q Healthc Ethics. 1993;2:505–515.
36. Meynen G. Depression, possibilities, and competence: a phenomenological perspective. Theor Med Bioeth. 2011;32:181–193.
37. Peternelj-Taylor AC. Conceptualizing nursing research with offenders: another look at vulnerability. Int J Law Psychiatry. 2005;28:348–359.
38. Fisher CB. Ethics in drug abuse and related HIV risk research. Appl Dev Sci. 2004;8:91–103.
39. Smebye KL, Kirkevold M, Engedal K. How do persons with dementia participate in decision making related to health and daily care? A multi-case study. BMC Health Serv Res. 2012;12:241.
40. Holm S. Autonomy, authenticity, or best interest: everyday decision making and persons with dementia. Med Health Care Philos. 2001;4:153–159.
41. Sabat SR. Capacity for decision making in Alzheimer’s disease: selfhood, positioning and semiotic people. Aust N Z J Psychiatry. 2005;39:1030–1035.
42. Kitwood T. Dementia Reconsidered: The Person Comes First. Birkshire: Open University Press; 1997.