
Innov Clin Neurosci. 2024;21(1–3):8–9.
Funding/financial disclosures. The authors have no conflicts of interest relevant to the content of this letter. No funding was received for the preparation of this letter.
Dear Editor:
Unexplained somatic symptoms are physical symptoms that cannot be explained by a known medical condition and are associated with autonomic imbalance. The estimated prevalence of unexplained somatic symptoms ranges from 20 to 74 percent in primary care settings and is about 50 percent in secondary care settings.1 Despite their frequent presentation in routine care, they are difficult to treat appropriately, reducing patient quality of life and placing an economic burden on society. Recent research indicated that maladaptive cognitions correlate with the severity of unexplained somatic symptoms, and, conversely, as somatic symptoms become more severe, maladaptive cognitions later appear.2 However, it is not known why unexplained somatic symptoms and maladaptive cognitions are closely related or how autonomic imbalances contribute to them.
Burning mouth syndrome (BMS) is a disorder included in the list of unexplained somatic symptoms. According to the International Classification of Headache Disorders, Third Edition (ICHD-3), BMS is defined as an oral burning sensation or dysesthesia that recurs daily for more than two hours, without a clinically evident causative lesion, lasting more than three months.3 Pain catastrophizing, one of the maladaptive cognitions, is characterized by the tendency to magnify the threat value of a pain stimulus and to feel helpless in the presence of pain, as well as by a relative inability to prevent or inhibit pain-related thoughts in anticipation of, during, or following a painful event. The QT interval has been noted as an indicator of autonomic imbalance. Therefore, we investigated the relationship between pain catastrophizing and QTc interval in patients with BMS. Patients with a diagnosis of BMS based on ICHD-3 criteria who made their first visit to our department without medication between April 2017 and April 2018 and whose consent was obtained were included in the study. A 12-lead electrocardiogram was performed at rest. Pain catastrophizing was assessed using the Pain Catastrophizing Scale (PCS), and a PCS score of 30 or higher indicated high pain catastrophizing.4 Data were analyzed using the Wilcoxon signed rank test.
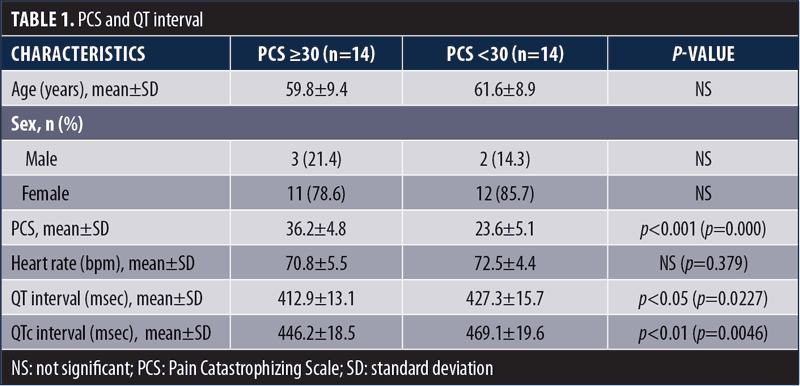
Results are shown in Table 1; the group with high pain catastrophizing had statistically, significantly shorter QT and QTc intervals than the group without high pain catastrophizing. Recent studies have shown that the QTc interval is associated with autonomic imbalances and has excellent sensitivity as a predictor of peripheral arterial disease and peripheral neuropathy in diabetes.5 In studies on healthy subjects, the QT interval indicated the degree of sympathovagal balance at a pulse rate of less than 100 per minute and tends to shorten with sympathetic tone.6 Thus, the results of this study suggest that pain catastrophizing is associated with sympathetic tone.
Brain functions are formed by functional connectivity between different neural regions, which are referred to as large-scale brain networks. The functional brain network consists of at least seven major networks: sensorimotor system, visual system, limbic system, dorsal attention network, central executive network, default mode network, and salience network. Studies using functional connectivity magnetic resonance imaging have shown that individuals with chronic pain have enhanced salience network activity, decreased functional connectivity between the default mode network and executive control network, and decreased functional connectivity between the default mode network and the descending pain inhibitory pathway.7 The salience network is strongly, functionally coupled to the dopaminergic reward system of the basal ganglia and increases sympathetic tone in the hypothalamus. Moreover, social distress, although not accompanied by physical pain, is associated with maladaptive cognitions, such as pathological loneliness or pain catastrophizing, and activates the cingulate gyrus and insular cortex, which make up the salience network, causing sympathetic tone.8 Maladaptive cognition might further enhance the salience network and sympathetic coupling seen in chronic pain, such as BMS. Thus, one possible mechanism for the bidirectional association between maladaptive cognition and unexplained somatic symptoms could be changes in the functional connectivity between large-scale brain networks and the autonomic nervous system.
Energy-intensive organs, such as the brain, attempt to conserve energy in every way possible. Acute pain, which rapidly increases sympathetic tone, increases daily energy expenditure by 60 percent.9 In contrast, chronic pain increases energy expenditure by only 15 percent.10 Chronic pain normally strengthens the functional connectivity between the somatosensory cortex and default mode network, increasing the parasympathetic tone and decreasing energy expenditure.11 However, pain catastrophizing in BMS might inhibit this process, resulting in increased functional coupling between the salience network and sympathetic nervous system.
The small number of cases, retrospective analysis, and the fact that sympathetic tone was assessed only by the QTc interval are limitations of this study. However, changes in functional connectivity between autonomic nervous system and large-scale brain networks that process pain might be involved in the vicious cycle of BMS symptoms and pain catastrophizing cognition. Further research is needed to prove this hypothesis.
References
- Zonneveld LN, Sprangers MA, Kooiman CG, et al. Patients with unexplained physical symptoms have poorer quality of life and higher costs than other patient groups: a cross-sectional study on burden. BMC Health Serv Res. 2013;13:520.
- Barends H, Dekker J, van Dessel NC, et al. Exploring maladaptive cognitions and behaviors as perpetuating factors in patients with persistent somatic symptoms: a longitudinal study. J Psychosom Res. 2023;170:111343.
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia. 2018;38(1):1–211.
- Darnall BD, Sturgeon JA, Cook KF, et al. Development and validation of a daily Pain Catastrophizing Scale. J Pain. 2017;18(9):1139–1149.
- Jiang AJ, Gu H, Feng ZR, et al. Heart rate-corrected QT interval: a novel diagnostic biomarker for diabetic peripheral neuropathy. J Diabetes Investig. 2022;13(5):850–857.
- Murakawa Y, Yamashita T, Ajiki K, et al. Is the QT interval an indicator of autonomic state? Jpn Heart J. 2000;41(6):713–721.
- De Ridder D, Vanneste S, Smith M, Adhia D. Pain and the triple network model. Front Neurol. 2022;13:757241.
- Eisenberger NI. The neural bases of social pain: evidence for shared representations with physical pain. Psychosom Med. 2012;74(2):126–135.
- Holland-Fischer P, Greisen J, Grøfte T, et al. Increased energy expenditure and glucose oxidation during acute nontraumatic skin pain in humans. Eur J Anaesthesiol. 2009;26(4):311–317.
- Straub RH. The brain and immune system prompt energy shortage in chronic inflammation and ageing. Nat Rev Rheumatol. 2017;13(12):743–751.
- Pei Y, Zhang Y, Zhu Y, et al. Hyperconnectivity and high temporal variability of the primary somatosensory cortex in low-back-related leg pain: an fMRI study of static and dynamic functional connectivity. J Pain Res. 2020;13:1665–1675.
With regards,
Takahiko Nagamine, MD, PhD, and Takeshi Watanabe, DDS, PhD
Dr. Nagamine is with Sunlight Brain Research Center in Hofu, Yamaguchi, Japan, and Department of Psychosomatic Dentistry, Graduate School of Medical and Dental Sciences, Tokyo Medical and Dental University in Tokyo, Japan. Dr. Watanabe is with Department of Preventive Medicine, Tokushima University in Tokushima, Japan.
