 by Rajashekar Reddy Yeruva, MD; Yafei Shang, MD; Ben Schoenbachler, MD; Sharon Nuss, MS; and Rif S El-Mallakh, MD
by Rajashekar Reddy Yeruva, MD; Yafei Shang, MD; Ben Schoenbachler, MD; Sharon Nuss, MS; and Rif S El-Mallakh, MD
Dr. Yeruva is with Baylor Scott and White Medical Center in Round Rock, Texas. Drs. Shang, Schoenbachler, El-Mallakh, and Sharon Nuss are with the University of Louisville School of Medicine in Louisville, Kentucky.
FUNDING: No funding was provided for this study.
DISCLOSURES: The author has no conflicts of interest relevant to the content of this article.
ABSTRACT: The pathogenesis of schizophrenia is rarely attributed to dysfunction of the cerebellum. However, coordination of mental processes might involve the cerebellum and a cortico-cerebellar-thalamic-cortical circuit (CCTCC) that can mediate that process has been proposed. We present the case of a 31-year-old male patient with a family history of psychosis who developed schizophrenia in association with a slow-growing glioblastoma at the left posterior cerebellar pontine angle. Of interest is that his psychosis became refractory after surgical removal of the tumor that had no motor deficit consequences, suggesting that the greater disruption of the CCTCC due to surgical excision might be related to the worsening psychosis. The case supports the hypothesis of cognitive dysmetria and psychosis.
Keywords: Schizophrenia, psychosis, cerebellar lesion, mental health, pathology, neuroanatomy
Innov Clin Neurosci. 2021;18(7–9):47-49
Schizophrenia is a particularly disabling illness due to its associated cognitive, social, and occupational functional impairments. It affects roughly one percent of the population. Its pathophysiology remains unknown, but neuroimaging has revealed early and progressive neural tissue loss most notable in the cortical tissues in temporal, prefrontal, and parietal areas, as well as in the diencephalon and basal ganglia with consequent ventricular enlargement.1,2 The cerebellum is only infrequently implicated, although there are neuroanatomical bases for considering the cerebellum in the pathophysiology of this illness.3,4
Case Presentation
A male patient in his 30s presented to the emergency department with new onset psychosis. Per the patient and his mother, the patient began hearing the “voice of God” two weeks previously and then began to become more spiritual, reporting a religious transformation, and praying naked on the floor. Neurologic examination was normal. Complete metabolic profile and thyroid stimulating hormone were normal, and imaging was not performed at this time. The patient was admitted to a psychiatric unit and started on lithium (900mg daily) and an antipsychotic (olanzapine 10mg daily, which was discontinued due to sedation, aripiprazole 10mg, which was discontinued due to gastrointestinal upset, and risperidone 4mg daily). Following discharge, the patient’s adherence was uncertain. Six months later, the patient was readmitted with similar symptoms after driving to West Virginia because the voice of God had told him to do that. In the interim, the patient had lost his job as an accountant and became unemployed. The patient was initiated on paliperidone 9mg daily and did reasonably well, although he lost a second job due to his psychotic symptoms.
Testing with Measurement and Treatment Research to Improve Cognition in Schizophrenia (MATRICS) Consensus Cognitive Battery (MCCB) revealed deficits in visual learning, attention/vigilance, and social cognition. The patient started with a new provider who ordered brain imaging due to the late onset of psychosis and the apparent lack of prodrome, which was not performed during the initial encounter a year previously. A slow growing glioblastoma (15.5 x 11.5mm in axial section) was discovered in the cerebellum at the left posterior cerebellar pontine angle (Figure 1).
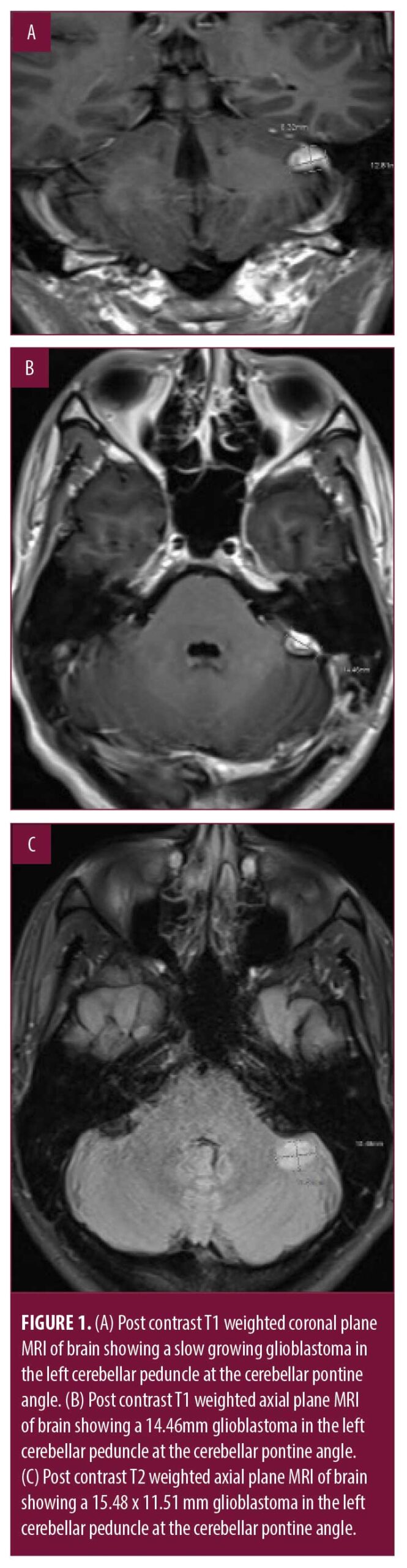
The assumpton was made that the psychotic symptoms were caused by the presence of the tumor and that removal of the tumor would improve the symptoms. We did not consider that removal of the tumor could actually increase the completeness of the lesion and make the psychosis worse. Four months later, the patient underwent a successful resection with no neurologic residual.
Soon after the surgery, the patient started experiencing continous auditory hallucinations of God, he was more religiously preoccupied, and he developed a delusion that he was married by God to a former coworker. Various medication changes and supratherapeutic antipsychotic doses (risperidone, ziprasidone, cariprazine) were ineffective despite apparent good adherence noted by family members.
Discussion
While this patient had an illness that met all criteria for schizophrenia, there were several aspects of his presentation that were unusual. There was no clear prodromal phase, and he was older (31 years old) than the more typical schizophrenia age of presentation.5,6 When the patient initially presented, imaging was not performed, so it is impossible to determine the temporal relationship between the cerebellar lesion and psychosis. However, it is clear the removal of the tumor, and the consequent possible additional disruption of the cortico-cerebellar-thalamic-cortical circuit (CCTCC), was associated with worsening of his symptoms.
The cerebellum has traditionally been seen as playing a role in motor regulation, modulation, and learning, but not as a major contributor to psychosis. Many early references to cerebellar influence on psychosis focused on the vermis,7–9 but recent studies have demonstrated hemispheric alteration in resting-state functional connectivity, including the left superior cerebellar peduncle,10,11 supporting potential involvement of left cerebellar hemispheric circuits. The cytostructure (or cytoarchitectonics) of the cerebellum is believed to enable it to recognize patterns, errors, and to modulate and coordinate cortical activity (both motor and cognitive).3,12 Dysfunction of these functions impairs synchrony, or the smooth coordination of mental processes. This process has been labeled cognitive dysmetria and defined as “difficulty in coordinating the processing, prioritization, retrieval, and expression of information” 13,14 Thus, “(i)nstead of modulating and coordinating, they . . . misconnect the billions of pieces of information arriving from the cerebral cortex. The output is then in turn flawed. For example, the location of an auditory signal arising from the auditory cortex without an external stimulus is misinterpreted as ‘outside’ rather than internal, leading to the experience of auditory hallucinations”.3 This process can lead to a range of diverse symptoms seen in schizophrenia.3,13 In our patient, we propose that as the tumor grew, it disrupted the CCTCC, producing the initial symptoms over the first year of his illness. The surgery removed the tumor with minimal motor sequelae but disrupted the CCTCC more fully, leading to a worsening of the psychosis.
This case provides indirect evidence of the role of the CCTCC in the development of psychotic symptoms. Additional data might help elucidate the role of this pathway in other patients with schizophrenia.
Take Home Points
Abnormal brain imaging occurs in less than five percent in patients with schizophrenia. Nonetheless,15,16 imaging is reasonable in the workup for schizophrenia if presentation is atypical.
Caution is indicated regarding assumptions that if symptoms are related to a lesion, then removal of the lesion would improve symptoms. In this case, removal of the lesion made the anatomical disruption more complete and was associated with worsening of the symptoms.
Cognitive dysmetria because of disruption of the CCTCC is a viable model for psychosis that might mimic schizophrenia.
References
- Weinberger DR, Torrey EF, Neophytides AN, et al. Lateral cerebral ventricular enlargement in chronic schizophrenia. Arch Gen Psychiatry. 1979; 36(7):735–739.
- Berger GE, Bartholomeusz CF, Wood SJ, et al. Ventricular volumes across stages of schizophrenia and other psychoses. Aust N Z J Psychiatry. 2017;51(10):1041–1051.
- Andreasen NC, Pierson R: The role of the cerebellum in schizophrenia. Biol Psychiatry. 2008;64(2):81–88.
- Mavroudis IA, Petrides F, Manani M, et al. Purkinje cells pathology in schizophrenia. A morphometric approach. Rom J Morphol Embryol. 2017;58(2):419–424.
- Malla AK, Norman RM. Prodromal symptoms in schizophrenia. Br J Psychiatry.
1994;164(4):487–493. - Eranti SV, MacCabe JH, Bundy H, et al. Gender difference in age at onset of schizophrenia: a meta-analysis. Psychol Med. 2013;43(1):155–167.
- Heath RG, Franklin DS, Shraberg D. Gross pathology of the cerebellum in patients diagnosed and treated as functional psychiatric disorders. J Nervous Mental Dis. 1979;167(10):589–592.
- Ichimiya T, Okubo Y, Suhara T, et al. Reduced volume of the cerebellar vermis in neuroleptic-naive schizophrenia. Biological Psychiatry. 2001;4:20–27.
- Garg S, Goyal N, Tikka SK, et al. Exacerbation of auditory verbal hallucinations with adjunctive high-frequency cerebellar vermal repetitive transcranial magnetic stimulation in schizophrenia: a case report. J ECT. 2013;29:65–66.
- Lee KH, Oh H, Suh JS, et al. Functional and structural connectivity of the cerebellar nuclei with the striatum and cerebral cortex in first-episode psychosis. J Neuropsychiatry Clin Neurosci. 2019;31(2):143–151.
- Zhuo C, Wang C, Wang L, et al. Altered resting-state functional connectivity of the cerebellum in schizophrenia. Brain Imaging Behav. 2018;12(2):383–389.
- Ito M. Cerebellar microcomplexes. Int Rev Neurobiol. 1997; 41:475–487.
- Andreasen NC, Nopoulos P, O’Leary DS, et al. Defining the phenotype of schizophrenia: cognitive dysmetria and its neural mechanisms. Biol Psychiatry.
1999;46:908–920. - Andreasen NC, Paradiso S, O’Leary DS.“Cognitive Dysmetria” as an integrative theory of schizophrenia: a dysfunction in cortical- subcortical-cerebellar circuitry? Schizophrenia Bull. 1998;24(2):203–218.
- Woolley J, McGuire P. Neuroimaging in schizophrenia: what does it tell the clinician? Adv Psychiatr Treat. 2005;11(3):195–202.
- Lubman DI, Velakoulis D, McGorry PD, et al. Incidental radiological findings on brain magnetic resonance imaging in first-episode psychosis and chronic schizophrenia. Acta Psychiatr Scand. 2002;106(5):331–336.
