by Richard G. Cockerill; Bridget K. Biggs, PhD; Tyler S. Oesterle, MD; and Paul E. Croarkin, DO
by Richard G. Cockerill; Bridget K. Biggs, PhD; Tyler S. Oesterle, MD; and Paul E. Croarkin, DO
Mr. Cockerill is a student at Mayo Medical School, College of Medicine, Mayo Clinic, Rochester, Minnesota; Drs. Biggs, Oesterle, and Croarkin are from Division of Child and Adolescent Psychiatry and Psychology, Mayo Clinic, Rochester, Minnesota.
Innov Clin Neurosci. 2014;11(11–12):14–21.
Funding: The Mayo School of Medicine provided funding for this study.
Financial disclosures: None of the authors have a conflict of interest in the conduct and reporting of this study.
Key words: Adolescent, Antidepressant, Depression, Obesity, SSRI
Abstract: Objective: Given the limited empirical data on antidepressant use and weight change in children, we performed a historical cohort study to assess change in age- and sex-standardized body mass index associated with antidepressant use among overweight adolescents diagnosed with a depressive disorder. Methods: We systematically reviewed electronic medical records from a tertiary academic medical center and identified adolescents (age 13–18 years) who were overweight (body mass index >85th percentile) and had a depression diagnosis. Patients were seen from January 1, 2000, through January 1, 2010. Age- and sex-standardized body mass index scores were calculated at initiation of antidepressant medication and at the end of treatment. Unmedicated patients had baseline and final age- and sex-standardized body mass index calculated using the first and last recorded measurements in the study period (maximum time between measures was 5 years). Results: In total, 435 patients (301 female) met our inclusion criteria; of these, 255 were prescribed an antidepressant (selective serotonin reuptake inhibitor, serotonin norepinephrine reuptake inhibitor, tricyclic antidepressant, or dopamine-norepinephrine reuptake inhibitor). Age- and sex-standardized body mass index significantly increased (F1,193=14.34; P<0.001) only for adolescents treated with selective serotonin reuptake inhibitors. For patients receiving other medications or no medication, age- and sex-standardized body mass index did not change significantly. Conclusion: This study provides initial empiric evidence for a link between selective serotonin reuptake inhibitor use and weight gain in already overweight adolescents. Further study of antidepressant use and weight gain in other pediatric populations and in prospective studies is warranted.
Introduction
Depression is a major public health problem that affects up to 11 percent of children in the United States.[1] Obesity is another major health concern among American youth. Approximately 12 percent of children (ages 2–9 years) have a body mass index (BMI) in the 97th percentile or greater.[2] Some studies suggest that obesity is more common among depressed children or adolescents.[3,4] Several, well-designed, prospective cohort studies have demonstrated an increased risk of obesity among children and adolescents with depression diagnoses at baseline,[5–7] but evidence that overweight youth are more likely to develop depression is lacking. Obesity can contribute to the development of depression over long periods[8] or among specific groups (e.g., non-Hispanic white girls),[9,10] yet no evidence has linked pre-existing overweight status or obesity to the development of depressive symptoms in adolescent populations.[11–14]
Antidepressants are frequently prescribed to treat depression throughout life. Some evidence suggests that antidepressants (e.g., mirtazapine, paroxetine, and tricyclic antidepressants [TCAs]) may be associated with weight gain in adults.[15] Selective serotonin reuptake inhibitors (SSRIs) often are considered weight neutral,[15,16] although some long-term studies have shown modest weight gain with fluoxetine and citalopram.[17,18] Moreover, recent evidence suggests that SSRIs increase the risk of metabolic disturbances and type II diabetes mellitus.[19–21] Bupropion, a dopamine and norepinephrine reuptake inhibitor, has been associated with weight loss in adults and has been used to treat psychotropic-induced weight gain.[15,22–24] The mechanisms underlying mirtazapine- and bupropion-induced weight change is not entirely clear. Mirtazapine is a strong antagonist of H1 histamine receptors, which can lead to increased appetite and weight gain.[25,26] This is also a mechanism by which atypical antipsychotics are thought to cause weight gain, along with their antidopaminergic effects.[27,28] Conversely, bupropion may promote weight loss by increasing the concentration of dopamine and norepinephrine in the brain, inducing a stimulatory and appetite-suppressing effect. However, these mechanisms are not well understood.[26,29]
A recent United States Centers for Disease Control and Prevention (CDC) report indicated that 3.7 percent of Americans aged 12 to 17 years were treated with an antidepressant from 2005 through 2008.[30] Little data are available on the association between antidepressant use and weight in overweight adolescents.[31] Because elevated BMI is a major public health issue for children and adolescents and because antidepressant treatment in adolescence is not uncommon, understanding the relation between antidepressants and BMI in this population is important, and understanding the impact of antidepressant treatment on obesity and overweight at-risk youth is critical.
To our knowledge, no prior study has examined whether antidepressant medications impact weight in overweight adolescents. Given the limited empirical data on antidepressant use and weight change in adolescent populations, this historical cohort study aimed to assess change in age- and sex-standardized BMI (zBMI) associated with antidepressant use among overweight adolescents diagnosed with a depressive disorder. Specifically, we anticipated that initiation of SSRIs and serotonin-norepinephrine reuptake inhibitors (SNRIs) would have little effect on zBMI and that zBMI change in these groups would be comparable to adolescents not using an antidepressant. Furthermore, we hypothesized that adolescents would have increased zBMI if treated with a tetracyclic antidepressant or decreased zBMI if treated with bupropion when compared with those who received no medication.
Method
This study was approved by our institutional review board. This was a retrospective review of patients whose parents or legal guardians authorized use of their medical records for research purposes, in accordance with state law.[32]
Subjects. We systematically searched electronic medical records to identify adolescent patients (age, 13–18 years), seen from January 1, 2000, through January 1, 2010, who met the diagnostic criteria for a depressive disorder (e.g., major depressive disorder [MDD], depression not otherwise specified, adjustment disorders, and dysthymia) and had a BMI at or above the 85th percentile for sex and age.[33]
Our rationale for studying overweight adolescents specifically was that weight-related side effects are particularly important in aiding providers in choosing antidepressants for this patient population, in light of the morbidity (e.g., metabolic syndrome, diabetes) associated with weight gain in already overweight patients. Adolescents who were prescribed an antidepressant had to have a trial of at least three months’ duration to be included in the analyses. Height and weight measures for all subjects had to be documented at least three months apart.
Patients were excluded if they had a diagnosis of an eating disorder, bipolar disorder, psychosis, schizophrenia, or schizoaffective disorder during the study period. Those with any medical condition known to affect BMI, including endocrine disorders (e.g., hypothyroidism, Cushing syndrome), gastrointestinal tract disorders (e.g., celiac disease, inflammatory bowel syndrome), any malignancy, and genetic disorders (e.g., Prader-Willi syndrome, Down syndrome) were also excluded. Furthermore, we excluded those who were prescribed atypical or typical antipsychotics, mood stabilizers (e.g., divalproex sodium, carbamazepine), stimulants, or atomoxetine.
Patients first were stratified by medication status (receiving medication or not). Patients who received medication were next stratified into five subgroups: SSRI, SNRI, TCA, tetracyclic antidepressants (e.g., mirtazapine), and bupropion. An a priori power analysis with G*Power 3.1.1[34] indicated that, with power set to 0.80 and alpha=0.05, a sample size of 140 was needed to detect the effects found in prior research on weight gain associated with mirtazapine, relative to placebo. Using the same analysis parameters, a sample size of 24 would be needed to detect weight loss found in prior research on bupropion.[35] We could not identify previously published data for weight change associated with SNRI, SSRI, or TCA use for the power analysis.
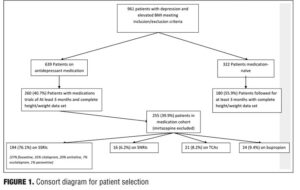
See Figure 1 for the consort diagram for patient selection.
Measures. From the medical records, we abstracted age, sex, height, weight, self-identified race or ethnicity, and antidepressant use at Time 1 and Time 2. Patient characteristics at initiation of antidepressant medication and at the end of treatment were recorded. For unmedicated patients, we used the first and last measurements recorded during the study period. We calculated BMI and zBMI[36] for each subject at both points and also recorded the time elapsed in months between measurements. Because of variation in the time between measurements among subjects, we normalized measures by calculating zBMI change per month.
Data analysis. SAS Enterprise 4.3 (SAS Institute, Inc.) was used for all analyses. Preliminary analyses examined medication class differences in initial age utilizing 2-way analysis of variance (ANOVA) with post-hoc Tukey HSD tests. Statistical significance was set at P Primary analysis. Five repeated-measures ANOVAs (1 for each medication class) were performed using Proc GLM, with zBMI at Time 1 and Time 2 as the repeated, within-subjects factor (time), tested hypotheses related to zBMI change within each medication class (i.e., whether zBMI significantly increased or decreased within each group). As a more stringent test of the hypotheses, we examined change in zBMI among the medication class relative to the no-medication group by performing a repeated-measures ANOVA using Proc GLM with zBMI at Times 1 and 2 as the repeated, within-subjects factor (time), medication as the between-subjects factor, and planned contrasts of each medication class with the no-medication group. Because of small subgroup sizes, sex and race or ethnicity were not included as between-subjects factors. Because age is accounted for in the calculation of zBMI, it was not needed as a covariate. Because of variation in the number of months elapsed between measurements, we then tested differences among medication classes in zBMI change per month using ANOVA with zBMI change per month as the dependent variable, medication as the between-subjects factor, and planned contrasts of each medication class with the no-medication group.
Results
Our initial search identified 961 patients. Of those who received antidepressant medication, 260 of 639 (40.7%) met criteria for the study, and of those who received no medication, 180 of 322 (55.9%) were included. The medication group was further divided into five classes: SSRI (n=194; 37% fluoxetine, 35% citalopram, 20% sertraline, 7% escitalopram, 1% paroxetine); SNRI (n=16; 100% venlafaxine), TCA (n=21), tetracyclic antidepressants (n=5; 100% mirtazapine), and bupropion (n=24). We decided to exclude the tetracyclic medication subgroup from the analyses because of low patient numbers. The final cohort consisted of 435 patients (301 female).
Patients primarily were white and not Hispanic/Latino (n=340). Eleven identified as African American, eight as Asian, one as Native Hawaiian/Pacific Islander, 30 as “other” race, and 28 as Hispanic/Latino. Race and ethnicity were recorded as “choose not to disclose” for one and two individuals, “unable to provide” for 19 and 15 individuals, and “unknown” for five and 17 individuals, respectively. Because of low racial diversity in the cohort, we used only two categories: 1) white, not Hispanic/Latino and 2) minority. Adolescents whose race was identified as black or African American, Asian, Native Hawaiian or Pacific Islander, or other or whose ethnicity was identified as Hispanic/Latino were included in the minority category. Those who indicated “unable to provide,” “unknown,” or “choose not to disclose” for race or ethnicity (n=36) were not included in the race and ethnicity analyses.
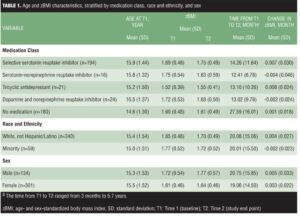
Preliminary analysis. Table 1 summarizes patient age and zBMI characteristics, stratified by medication class, race and ethnicity, and sex.
Differences in patient characteristics by medication Class. The medication classes differed significantly in initial age (F4,430=27.30; P<0.001). Specifically, the no-medication group was younger than all other groups except the TCA group, and the TCA group was younger than the bupropion group. The percentage of male and female subjects differed significantly by medication class (chi-squared4 =11.77; P=0.02; n=435); female subjects accounted for 69.2 percent of the total sample but were overrepresented in the TCA (81.0%), bupropion (83.3%), and SSRI (74.2%) groups, whereas male subjects were overrepresented in the no-medication group (38.9% vs 30.8% in the total sample). Distribution of race or ethnicity was not significantly different among the medication classes (chi-squared4 =4.46; P=0.35; n=435).
zBMI by sex and race or ethnicity. Female subjects had lower zBMI scores than did male subjects at Time 1 (F1,434=4.60; P=0.03) and at Time 2 (t434=6.33; P=0.01), but we observed no significant time-x-sex interaction (F1,433=0.46; P=.50), which indicated that males and females did not differ in zBMI change. Further, a t test comparing females and males on rate of zBMI change was not significant (t434=0.99; P=0.32). There were no significant differences in minority status for zBMI at Time 1 (t398=3.12; P=0.08) or Time 2 (t398=0.09; P=0.77), but the time-x-minority status interaction was significant (F1,397=5.83; P=0.02). Follow-up repeated-measure ANOVAs run separately by minority status group indicated that zBMI increased significantly among white, not-Hispanic/Latino adolescents (F1,339=7.95; P=0.005) but not among minority adolescents (F1,58=2.26; P=0.14). However, a t-test of minority status differences in the rate of zBMI change was not significant (t398=2.64; P=0.10).
Primary analyses. In the repeated-measures ANOVAs examining zBMI change within each medication class, the effect for time was significant only for the SSRI group (F1,193=14.34; P<0.001). The effect was not significant for the SNRI (F1,15=1.19; P=0.29), TCA (F1,20=0.26; P=0.62), bupropion (F1,23=0.33; P=0.57), and no-medication (F1,179=0.14; P=0.71) groups.
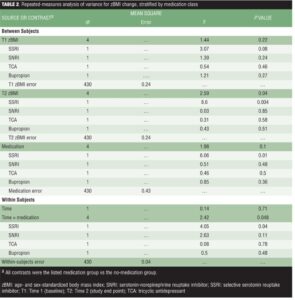
The repeated-measures ANOVA for zBMI change by medication class relative to the no-medication group showed no significant differences in zBMI across medication classes at Time 1 (i.e., no main effect for medication at Time 1), and none of the specific contrasts for Time 1 zBMI were significant (Table 2).
However, we observed significant differences across medication classes in Time 2 zBMI (i.e., a significant main effect for medication at Time 2). The planned contrasts indicated significantly greater zBMI at Time 2 only in the SSRI group relative to the no-medication group. Importantly, results indicated a significant time-x-medication interaction effect such that zBMI increased in the SSRI group relative to the no-medication group.
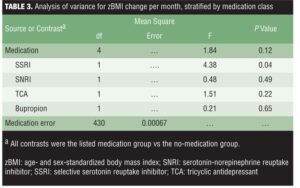
When examining medication class differences in zBMI change per month, results were consistent with the repeated-measures ANOVA (Table 3). Specifically, only the contrast comparing the SSRI group with the no-medication group was significant, indicating a greater increase in zBMI per month in the SSRI group.
Discussion
This study contributes initial data regarding zBMI change associated with treatment with antidepressant medications among overweight adolescents. Our cohort was consistent with previously observed higher prevalence of depressive disorders among female relative to male adolescents.[37] Contrary to previous studies suggesting that SSRIs are weight neutral in adults,[15,38] the current study showed zBMI increasing among depressed and overweight adolescents who took SSRIs versus those received no medication. This finding underscores the need for further study of the effects of this medication class in pediatric patients, particularly among those who are already overweight. SSRIs were the most commonly prescribed antidepressant in this retrospective study. This was expected, given that it is considered one of the best practices for the treatment of child and adolescent depression.[39]
We did not observe a significant zBMI change associated with SNRIs; however, this observation should be interpreted with caution because only 16 patients were prescribed this class of medication during the 10-year study period. SNRIs are not currently approved by the United States Food and Drug Administration (FDA) for adolescents and are not considered a standard pharmacologic treatment by child and adolescent psychiatrists.[40] Contrary to our hypotheses, we did not observe a significant zBMI decrease among the bupropion group, despite having a sufficiently large sample to detect the degree of weight loss observed in previous studies.[23,24] The number of adolescents prescribed a tetracyclic medication (n=5) was insufficient to test the hypothesis that this class would be associated with a zBMI increase.
Interestingly, an incidental finding from our preliminary analyses indicated that white, not-Hispanic/Latino adolescents, overall, had an increase in zBMI during the study period, whereas those from minority groups did not. The observed weight gain among white adolescents was consistent with prior research demonstrating weight gain among individuals with depression,[5-7] but the observation of weight stability among racial and ethnic minority adolescents is unexpected, considering the greater prevalence of pediatric obesity in these groups, particularly black and Latino youth.[2] Small sample sizes prevented the examination of zBMI and zBMI change among specific minority groups and the investigation of zBMI change by medication class and minority status. Larger and more representative samples are needed to further examine the effects of antidepressant use among racial and ethnic groups. Given the role of developmental risk factors in racial or ethnic differences in childhood obesity, further study with specific minority groups is crucial.[41]
It is premature to caution against SSRI use among overweight adolescents, particularly because this class of medications is currently considered an effective treatment option for pediatric depression and includes the only FDA-approved medications for major depressive disorder in adolescents (fluoxetine and escitalopram).[42,43] Available empirical information on weight gain or loss and other potential adverse effects needs to be considered along with evidence of the effectiveness of available treatments, patient characteristics, and patient preferences.
Limitations. Important limitations to the current study are that it was retrospective and did not include random assignment, which limited our ability to control for historical confounders that may have influenced practitioner prescribing practices (i.e., group assignment) or zBMI change in some groups more than others. To illustrate, it is possible that adolescents with more severe depression were more likely to be prescribed an antidepressant (typically an SSRI). If adolescents with more severe depression struggled more with eating well and being physically active, the weight gain among the SSRI group would be due to greater depression severity rather than the medication itself. Alternatively, if prescribers considered patient factors related to risk for weight gain in selecting a medication (or opting for no medication), such factors could have influenced the study results. For example, prescribers may have selected SSRIs for adolescents they saw as at risk for further weight gain, with the assumption that these agents were weight neutral. The current study, being retrospective, could not measure these variables.
Another key limitation secondary to study design was that we could not monitor medication adherence during follow-up. This may confound conclusions made regarding the association found between weight increase and SSRI use.
Future prospective studies that include random assignment and/or measurement of potential confounding variables would address these issues and bring greater clarity to the current findings. Further, future prospective studies could include potential mediators such as eating habits, physical activity, parental BMI, depression severity, and inflammatory markers such as C-reactive protein, absolute neutrophil count, and ferritin/transferrin ratios.[44] Such studies have the potential to shed light on mechanisms underlying observed zBMI changes or stability among adolescents taking various classes of antidepressants or receiving no medication.
Although power estimates established before the chart review indicated that the sample size of SSRIs and bupropion were sufficient to detect zBMI changes in these groups relative to the no-medication group, the study power to detect differences in zBMI change in the other medication classes relative to the no-medication group may have been limited. Studies with larger samples of adolescents prescribed SNRIs, tricyclics, and tetracyclics are needed to determine whether they are associated with weight change in overweight adolescents. However, this could prove challenging for pragmatic and ethical reasons. The low rates of use in the present study may reflect good clinical practice because SSRIs have stronger evidence of effectiveness for depression and tricyclic antidepressants are associated with weight gain in adults.[45]
Conclusion
This is the first study that, to our knowledge, examines weight gain among depressed, overweight adolescents receiving pharmacologic treatment for depression. We present preliminary evidence of a link between SSRI use and weight gain in these overweight youth. Future prospective studies should consider additional confounders and examine potential biologic mechanisms.
Acknowledgment
Statistical analysis was conducted by one of the authors, Dr. Bridget K. Biggs.
References
1. Merikangas KR, He JP, Burstein M, et al. Lifetime prevalence of mental disorders in U.S. adolescents: results from the National Comorbidity Survey Replication—Adolescent Supplement (NCS-A). J Am Acad Child Adolesc Psychiatry. 2010;49(10):980–989. Epub 2010 Jul 31.
2. Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999–2010. JAMA. 2012;307(5):483–490. Epub 2012 Jan 17.
3. Phillips BA, Gaudette S, McCracken A, et al. Psychosocial functioning in children and adolescents with extreme obesity. J Clin Psychol Med Settings. 2012;19(3):277–284.
4. Gracious BL, Cook SR, Meyer AE, Chirieac MC, Malhi N, Fischetti AT, et al. Prevalence of overweight and obesity in adolescents with severe mental illness: a cross-sectional chart review. J Clin Psychiatry. 2010 Jul;71(7):949–54. Epub 2010 May 4.
5. Anderson SE, Cohen P, Naumova EN, Must A. Association of depression and anxiety disorders with weight change in a prospective community-based study of children followed up into adulthood. Arch Pediatr Adolesc Med. 2006;160(3):285–291.
6. Goodman E, Whitaker RC. A prospective study of the role of depression in the development and persistence of adolescent obesity. Pediatrics. 2002;110(3):497–504.
7. Pine DS, Goldstein RB, Wolk S, Weissman MM. The association between childhood depression and adulthood body mass index. Pediatrics. 2001;107(5):1049–1056.
8. Mustillo S, Worthman C, Erkanli A, et al. Obesity and psychiatric disorder: developmental trajectories. Pediatrics. 2003;111(4 Pt 1):851–859.
9. Goodman E, Must A. Depressive symptoms in severely obese compared with normal weight adolescents: results from a community-based longitudinal study. J Adolesc Health. 2011;49(1):64–69. Epub 2011 Mar 12.
10. Erickson SJ, Robinson TN, Haydel KF, Killen JD. Are overweight children unhappy? Body mass index, depressive symptoms, and overweight concerns in elementary school children. Arch Pediatr Adolesc Med. 2000;154(9):931–935.
11. Britz B, Siegfried W, Ziegler A, et al. Rates of psychiatric disorders in a clinical study group of adolescents with extreme obesity and in obese adolescents ascertained via a population based study. Int J Obes Relat Metab Disord. 2000;24(12):1707–1714.
12. Friedman MA, Wilfley DE, Pike KM, et al. The relationship between weight and psychological functioning among adolescent girls. Obes Res. 1995;3(1):57–62.
13. Lamertz CM, Jacobi C, Yassouridis A, et al. Are obese adolescents and young adults at higher risk for mental disorders?: a community survey. Obes Res. 2002;10(11):1152–1160.
14. Wardle J, Williamson S, Johnson F, Edwards C. Depression in adolescent obesity: cultural moderators of the association between obesity and depressive symptoms. Int J Obes (Lond). 2006;30(4):634–643.
15. Serretti A, Mandelli L. Antidepressants and body weight: a comprehensive review and meta-analysis. J Clin Psychiatry. 2010;71(10):1259–1272.
16. Schwartz TL, Nihalani N, Jindal S, et al. Psychiatric medication-induced obesity: a review. Obes Rev. 2004;5(2):115–121.
17. Ribeiro L, Busnello JV, Kauer-Sant’Anna M, et al. Mirtazapine versus fluoxetine in the treatment of panic disorder. Braz J Med Biol Res. 2001;34(10):1303–1307.
18. Leinonen E, Skarstein J, Behnke K, et al, Nordic Antidepressant Study Group. Efficacy and tolerability of mirtazapine versus citalopram: a double-blind, randomized study in patients with major depressive disorder. Int Clin Psychopharmacol. 1999;14(6):329–337.
19. Andershon F, Schade R, Suissa S, Garbe E. Long-term use of antidepressants for depressive disorders and risk of diabetes mellitus. Am J Psychiatry. 2009;166(5):591–598
20. Jerrell JM, Tripathi A, Rizvi AA, McIntyre RS. The risk of developing type 2 diabetes mellitus associated with psychotropic drug use in children and adolescents: a retrospective cohort analysis. Prim Care Companion CNS Disord. 2012;14(1).
21. Khoza S, Barner JC, Bohman TMS, et al. Use of antidepressant agents and the risk of type 2 diabetes. Eur J Clin Pharmacol. 2012;68(9):1295–1302.
22. Levine MD, Perkins KA, Kalarchian MA, et al. Bupropion and cognitive behavioral therapy for weight-concerned women smokers. Arch Intern Med. 2010;170(6):543–550.
23. Gadde KM, Zhang W, Foust MS. Bupropion treatment of olanzapine-associated weight gain: an open-label, prospective trial. J Clin Psychopharmacol. 2006;26(4):409–413.
24. Wadden TA, Foreyt JP, Foster GD, et al. Weight loss with naltrexone SR/bupropion SR combination therapy as an adjunct to behavior modification: the COR-BMOD trial. Obesity (Silver Spring). 2011;19(1):110–120. Epub 2010 Jun 17.
25. Anttila SA, Leinonen EV. A review of the pharmacological and clinical profile of irtazapine. CNS Drug Rev. 2001;7(3):249–264.
26. Feighner JP. Mechanism of action of antidepressant medications. J Clin Psychiatry. 1999;60 Suppl 4:4–11.
27. Kroeze WK, Hufeisen SJ, Popadak BA, et al. H1-histamine receptor affinity predicts short-term weight gain for typical and atypical antipsychotic drugs. Neuropsychopharmacology. 2003;28(3):519–526.
28. Correll CU, Manu P, Olshanskiy V, et al. Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. JAMA. 2009;302(16):1765–1773. Erratum in: JAMA. 2009;302(21):2322.
29. Plodkowski RA, Nguyen Q, Sundaram U, et al. Bupropion and naltrexone: a review of their use individually and in combination for the treatment of obesity. Expert Opin Pharmacother. 2001;10(6):1069–1081.
30. Pratt LA, Brody DJ, Gu Q. Antidepressant use in persons aged 12 and over: United States, 2005–2008 [internet]. Number 76, October 2011. Atlanta (GA): Center for Disease Control and Prevention. http://www.cdc.gov/nchs/data/databriefs/db76.htm. Accessed December 18, 2014.
31. Jerrell JM. Neuroendocrine-related adverse events associated with antidepressant treatment in children and adolescents. CNS Neurosci Ther. 2010;16(2):83–90. Epub 2009 Sep 21.
32. Disclosure of health records for external research. Minn Rev Stat. § 144.295 (2012).
33. Barlow SE, Expert Committee. Expert committee recommendations regarding the prevention, assessment, and treatment of child and adolescent overweight and obesity: summary report. Pediatrics. 2007;120 Suppl 4:S164–92.
34. Download and register G*Power 3 [Internet]. Dusseldorf (Germany): Heinrich Heine Universitat Dusseldorf [German]. http://www.psycho.uni-duesseldorf.de/abteilungen/aap/gpower3/download-and-register. Accessed December 18, 2014.
35. Gadde KM, Parker CB, Maner LG, et al. Bupropion for weight loss: an investigation of efficacy and tolerability in overweight and obese women. Obes Res. 2001;9(9):544–551.
36. A SAS program for the CDC growth charts [internet]. Atlanta (GA): Centers for Disease Control and Prevention. January 13, 2014 http://www.cdc.gov/nccdphp/dnpao/growthcharts/resources/sas.htm. Accessed December 18, 2014.
37. Hankin BL, Abramson LY, Moffitt TE, et al. Development of depression from preadolescence to young adulthood: emerging gender differences in a 10-year longitudinal study. J Abnorm Psychol. 1998;107(1):128–140.
38. Schwartz TL, Nihalani N, Virk S, et al. Psychiatric medication-induced obesity: treatment options. Obes Rev. 2004;5(4):233–238.
39. Birmaher B, Brent D, AACAP Work Group on Quality Issues. Practice parameter for the assessment and treatment of children and adolescents with depressive disorders. J Am Acad Child Adolesc Psychiatry. 2007;46(11):1503–1526.
40. Pfalzgraf AR, Scott V, Makela E, et al. Child psychiatrists’ self-reported treatment and monitoring of children and adolescents with major depressive disorder. J Psychiatr Pract. 2012;18(4):253–261.
41. Taveras EM, Gillman MW, Kleinman KP, et al. Reducing racial/ethnic disparities in childhood obesity: the role of early life risk factors. JAMA Pediatr. 2013;167(8):731–738.
42. Correll CU, Kratochvil CJ, March JS. Developments in pediatric psychopharmacology: focus on stimulants, antidepressants, and antipsychotics. J Clin Psychiatry. 2011;72(5):655–670.
43. Sakolsky D, Birmaher B. Developmentally informed pharmacotherapy for child and adolescent depressive disorders. Child Adolesc Psychiatr Clin N Am. 2012;21(2):313–25. Epub 2012 Feb 24.
44. Skinner AC, Steiner MJ, Henderson FW, Perrin EM. Multiple markers of inflammation and weight status: cross-sectional analyses throughout childhood. Pediatrics. 2010;125(4):e801–9. Epub 2010 Mar 1.
45. Leucht C, Huhn M, Leucht S. Amitriptyline versus placebo for major depressive disorder. Cochrane Database Syst Rev. 2012;12:CD009138.