
by Franco Di Cesare, MD; Cristiana Di Carlo, MSc; and Leonardo Di Cesare, MD
Drs. F. Di Cesare and Di Carlo are with Leoben Research srl in Rome, Italy. Dr. L. Di Cesare is with Glaxo-Smith-Kline in Siena, Italy.
Funding: The project was sponsored and funded by Leoben Research.
Disclosures: Drs. F. Di Cesare and Di Carlo are co-founders and co-owners of Leoben Research. Dr. L. Di Cesare was previously employed by Leoben Research.
Innov Clin Neurosci. 2023;20(1–3):46–52.
Abstract
Objective: The assessment of child cognitive health in Sub-Saharan developing countries poses significant challenges, including the paucity of valid diagnostic tools. We report the development and the initial validation of the Zambia Symbol Cancellation Test (ZSCT), a psychometric test to evaluate selective attention in a pediatric, school-aged population.
Design: ZSCT reliability and validity were assessed in a field trial. A total of 409 children, aged 5 to 17 years, were recruited at multiple Zambian clinical sites and schools. The ZSCT provides a visuomotor processing index (VMPI), a measure of effortful processing to deliver accurate task response.
Results: The VMPI reliability estimate at test-retest was found to be adequate for a clinical use (intraclass correlation coefficient [ICC]: 0.79, ICC-95% confidence interval [CI]: 0.69–0.86). Age showed a large effect on VMPI (n=323, r=-0.62, p=0.000). Impaired visuo-perceptual-motor processing, as measured by VMPI, was associated with poor health status (i.e., chronic neurologic or medical disease or prolonged exposure to psychosocial stress and deprivation). A two-way ANOVA found significant and small health status and age group effects [F (7, 408): 33.24, p=0.0000, η2=0.367]; the main effect of health status [F (1, 408): 37.79, p=0.000, η2=0.09], age group [F (3, 408): 35.06, p=0.000, η2=0.21], and their interaction was not significant (p=0.364).
Conclusion: Study findings indicate that the ZSCT has satisfactory reliability, validity, and clinical utility to evaluate cognitive development and the effect of health conditions on attention. Study results warrant further research to validate its use in healthcare and clinical research settings.
Keywords: Clinical trials, pediatric cognitive assessment, attention, visuomotor processing, test development, developing countries
Attention is a cognitive domain of particular interest in pediatric cognitive assessment due to its importance as an indicator of a child’s health status. However, the scarce availability of valid diagnostic instruments is one of the barriers preventing a satisfactory assessment of cognitive outcomes at individual (e.g., child in a healthcare setting), group (e.g., treatment group in an experimental design), or population levels in Sub-Saharan African countries.1
Zambia is an example of a Sub-Saharan developing country where the unmet medical need of more cognitive assessment tools, including ones to evaluate attention, is evident. The Zambian population mostly relies on a small number of diagnostic instruments that were originally developed in North American and European countries, then imported to Zambia for use in healthcare and research settings.2,3 For example, the assessment of attention in school-aged children (i.e., aged between 5 and 17 years) is limited to subtests of the Developmental Neuropsychological Assessment (NEPSY).2,4,5 NEPSY is a test battery originally developed and normed on a pediatric population in a high-income country (United States [US]) to address specific assessment needs in that socioeconomic and cultural context, which is very different from the socioeconomic and cultural context of Zambia. Nevertheless, the use of diagnostic instruments developed and standardized in a different cultural context carries the risk of poor cross-cultural validity, and, ultimately, misrepresenting the cognitive abilities of children.2,3,6 To date, notable attempts to develop a Zambian-culture appropriate test to evaluate cognition, such as Panga Munthu;7,8 Cognitive Ability Assessment Tool;9 Zambia Chlorhexidine Application Trial (ZamCAT), a test battery for multiple neuro-cognitive domains for children aged 5–6 years;10 and Object-based Pattern Reasoning Assessment,11 do not measure attention in children aged 5 to 17 years.
Attention and cognitive development. Attentive behavior is characterized by a conscious, intentional, and focused effort to maintain efficient cognitive processing of a specific, goal-oriented task.12 Attention underlies the behavioral ability to attend to some events while ignoring others12 and works as a selective filter, actively choosing some information for extensive processing at the expense of others.12–15 Attention is key to prioritizing available information for more in-depth coding and integration, as processing of all information would be overwhelming, unnecessary, and counterproductive.
Attentive behavior is also conceptualized as the outcome of a unified neurobehavioral functional system for regulation and control of cognitive processing.16 Attentive behavior corresponds to the activity of a network of functionally integrated anatomical brain areas, wherein different networks can specify for diverse modalities of cognitive processing underlying a specific attentive behavior;16 therefore, functional attention subdomains within the unitary system can be constructed and assessed using specific behavioral paradigms.
Selective attention is a functional subdomain aimed at overcoming the limited capacity of the cognitive system to process information. Selective attention is characterized by cognitive processing of relevant and specific targets of information (e.g., perceptual stimuli, ideations, and behaviors) and simultaneous neglect or attenuation of other sources of irrelevant information when engaging in a goal-directed task.13–17 Selective attention is distinct from focused attention, in which cognitive processing aims at the identification and signaling of targets only and not of distractors; sustained attention, which involves maintenance of cognitive processing on a continuous and repetitive performance task over a prolonged time interval (i.e., many minutes); and divided attention, which requires cognitive processing as attending multiple tasks in parallel.
Selective attention plays a unique role in connecting the behavioral level of description of cognitive processing (i.e., a specific attentive behavior) with brain developmental and functional anatomy and neurobiology. The neural systems functionally associated with selective attention, including regions of the prefrontal and parietal cortices, show a prolonged period of postnatal structural development throughout childhood, adolescence, and early adulthood. The slow maturation of the basic neural substrates may occur in parallel with functional changes in selective attention.18 Furthermore, health conditions, pharmacological effects, and other factors can result in altered brain functioning and impair selective attention.19–21
Measuring selective attention in a school-aged Sub-Saharan population. The measurement of selective attention can be applied to address specific knowledge needs in pediatric clinical healthcare and research settings, such as by providing information on a child’s cognitive development. A deficit of selective attention in a child impacts very early stages of perceptual processing and has cascading consequences on the development of other cognitive abilities, including numeracy, language, and reading.18,22 A subsequent, broader cognitive impairment has the potential to deter the normal development of the child and poses a threat to full functioning in life. The conceptual framework of cognitive development as a dynamic interplay between age and biological and environmental factors suggests that cognitive abilities can be enhanced by interventions that rebalance protective and health-risk factors.
Early detection and precise definition of selective attention deficit can identify children who are at increased risk for altered cognitive development and provide insight on the most appropriate measures to adopt for clinical management. This modality of intervention can result in better health and psychosocial outcomes.22 Furthermore, assessment of selective attention in clinical and epidemiological research help to better characterize the clinical manifestations and burden of medical or psychosocial conditions on childhood cognitive functioning and development.1,19 Finally, measures of selective attention can also be applied to pediatric clinical trial design for the purpose of diagnostic classification or as evaluation outcomes of intervention programs.1,23
Developing a selective attention test in a Sub-Saharan school-aged population. A development strategy for a new method of assessing selective attention for use in Sub-Saharan developing countries should be based on an original design of a culturally appropriate tool and its validation in the specific context of use.
The attentive behavior chosen for testing should be familiar to the child, relevant to the child’s everyday life experience, and frequently occurring in a pediatric population. An optimal design of measures of selective attention for healthcare and research applications should also satisfactorily address the issues of a user-friendly and simplified procedure of test administration, streamlined scoring procedure, and limited time resource for assessment. Target cancellation tasks satisfy these criteria. A target cancellation task involves visuomotor searching for detection and signaling of a predefined target against a background of distractors that are spatially arranged (e.g., in rows and columns). In a typical paper-and-pencil target cancellation task, a sheet of paper is placed directly in front of the child showing random arrays of stimuli, some of them being targets to be signaled by marking (hits) and the remaining ones to be skipped (nontarget distractors).
Target cancellation tasks have been widely used in experimental and clinical settings to investigate attention deficits in children.20,24–27 Typically, outcomes include the number of correctly signaled targets (alternatively, omitted targets), the number of erroneously marked nontargets, and task processing time.25,28 There is a relationship between target cancellation performance and age, whereby accuracy of response increases with age in typically developing children.20,27,29,30 Changes in attention outcomes are more evident in early and late childhood and stabilize during adolescence.29,30
Purpose of the present research. To the best of our knowledge, no test to evaluate selective attention in Zambian children is currently available. A multidisciplinary group of Zambian and international field experts, the Zambia Paediatric Cognitive Assessment Working Group, conducted a research program on new diagnostic instruments for a school-aged population in Sub-Saharan countries. We report the development and initial validation of the Zambia Symbol Cancellation Test (ZSCT). A field trial was conducted to evaluate psychometric properties of the ZSCT in a Zambian school-aged population. We assessed construct validity; reliability, defined as stability of a repeated measurement; and potential for clinical utility to evaluate attention deficits
Materials and Methods
Instrument. The ZSCT is based on the administration of a speeded and continuous target cancellation task. The data capture sheet (Figure 1) is directly placed in front of the child showing an organized, monochromatic array (dimensions: 25cm x 17cm) containing 324 familiar black symbols (*, <, + ), 108 targets and 216 distractors, equally distributed across the four quadrants, with 27 targets and 54 distractors in each quadrant. The child is asked to indicate the target of the task by selecting only one of the three symbols. Then, the child is given the instruction to mark every corresponding target with a single slanted line as fast as they can until the task is completed. The ZSCT is a speeded task, as it is performed under a time constraint of 180 seconds for completion. Three outcomes are noted: sum of hits, sum of commissions, and task processing time in seconds. A hit is defined as a correctly signaled target; there are 108 targets (i.e., the symbol chosen by the child) to hit, 27 in each quadrant. Commission is an incorrectly signaled target; the maximum number of commissions is 216, 54 in each quadrant.

The structure of the cancellation task satisfies key requirements to test the construct of selective attention. First, the task allows for selective attention to occur (i.e., the child can exercise selective attention in full through inhibition of response to distractors and focusing on targets to hit simultaneously). Second, it provides measurement outcomes for both the degree to which a child inhibits the response to distractors (i.e., frequency of occurrence of commissions provides a quantitative outcome of response to distractors) and to which a child attends to the targets (i.e., frequency of hits). The task involves a conscious, intentional, and continuous effort to maintain the efficient management of visuo-perceptual-motor processing of a specific, goal-oriented task (i.e., signaling of true targets as avoiding distractors). Finally, the time constraint is applied to maximize the cognitive demand for resourcing and gradient of attentional capabilities during the cancellation task. A time limit of 180 seconds makes the ZSCT unable to test sustained attention that requires a continuous performance task lasting several minutes.
Design. This study was a field cross-sectional trial combined with an ancillary test-retest reliability study on a selected subgroup of study participants.
Ethical considerations. We conducted the trial in accordance with Zambian legal and regulatory requirements. The study obtained approval from the National Health Research Authority and the University of Zambia Biomedical Research Ethics Committee (reference number 008-08-165, approval on February 27, 2018). Parents or legal guardians of children participating in this study signed a written, informed consent form, including assent from the child if aged 12 years or older. The informed consent process was carried out in the presence of an independent witness for illiterate parents or guardians.
Participants. To be enrolled in the study, participants had to be 5 to 17 years of age (including limits), have access to formal education, be experienced and familiar with cognitive tasks similar to ones required by the ZSCT (e.g., written marking), be able to communicate effectively with the examiner, and provide parental consent for participation.
Exclusion criteria included presence of a serious health condition currently requiring inpatient hospitalization, significant visuoperceptual disability not corrected by glasses or lenses, significant speech disability, hearing impairments, significant sensory or motor disabilities, current acute medical or mental health condition, any significant chronic conditions that prevented the execution of cognitive tests, inability to understand test instructions, or being uncooperative during the execution of the clinical assessments.
Selected participants and the sites for test administration represented a broad sample of children and settings in which the ZSCT is intended for use. The study population was recruited from multiple clinical and school sites in two diverse regions of Zambia, the Lusaka Metropolitan Area and the Copperbelt Province. Schools in Lusaka and the Copperbelt Province were purposely selected to provide a heterogeneous sample, representative of the wide socioeconomic, cultural, and linguistic diversity of the Zambian pediatric population. Clinical sites were selected to ensure that a heterogeneous sample, representative of the diversity of medical and psychosocial conditions affecting child health and cognitive development, was drawn. Clinical sites were outpatient clinics at the University Teaching Hospital Department of Pediatrics and Child Health in Lusaka and tertiary outpatient clinics in the Copperbelt Province, Ndola area. University Teaching Hospital is the highest referral hospital in Zambia, delivering specialized care in pediatrics, and receives children from all over the country. Enrollment started in August 2018 and ended in October 2019.
Data collection methods and analysis. Sociodemographic and medical history information was collected, and the ZSCT was administered to all study participants. A subsample of participants also underwent clinical and neurological examinations and electroencephalographic (EEG) evaluation.
STATA software version 15.1 was used for all statistical analyses.31 Parametric and nonparametric tests, linear regression model, and ANOVA were applied to evaluate the effect of sex, age, and health status on ZSCT outcomes.
Construct validity was tested using a multiple linear regression model to predict sum of hits based on task processing time as the predictor.
Reliability, as stability of the measure over time, was evaluated applying a single-measurement, absolute agreement, two-way, mixed-effects model.32 Coefficients of intraclass correlations (ICs) were calculated as point estimates. Two-way mixed-effects method was applied because repeated measurements cannot be assumed as randomized samples.
Results
The evaluation of psychometric properties was conducted in a sample of 323 school-aged children (Table 1). The sample included children in apparent good health at the time of assessment, although they may have had a chronic health condition that did not represent an important risk or causative factor for cognitive impairment. They all had access to formal education and regularly attended school. All children were from an urban area.
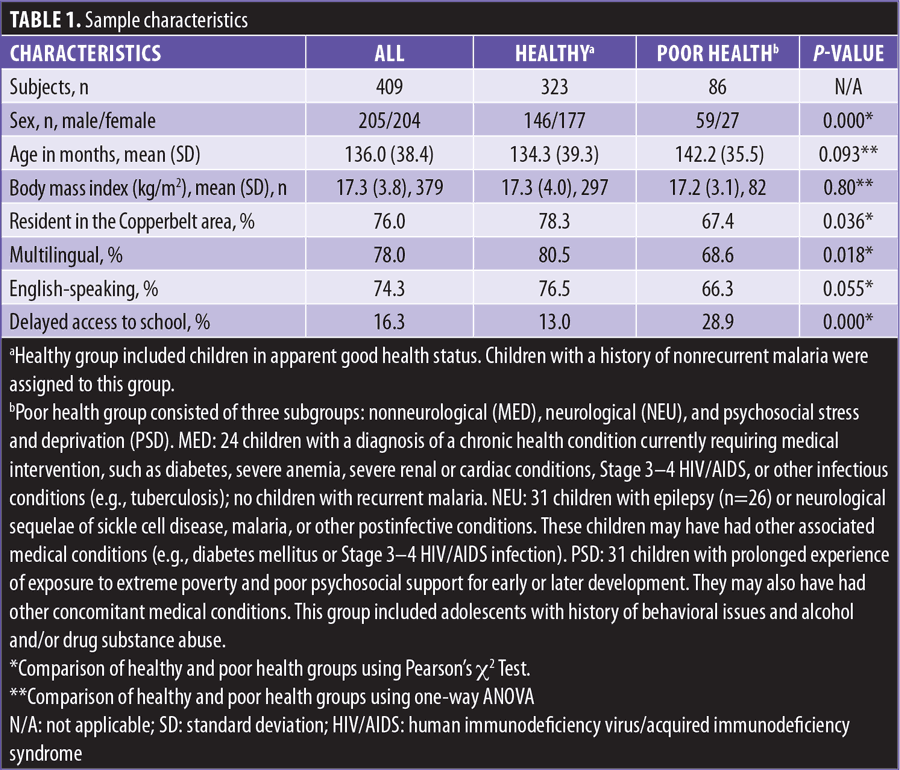
Summary statistics of ZSCT outcomes are shown in Table 2. Prevalence of commissions was relatively low. We found a large effect of age on task processing time (n=323, r= -0.58, p=0.000) and sum of hits (n=323, r=0.50, p=0.000). There was no statistically significant correlation between age and sum of commissions.
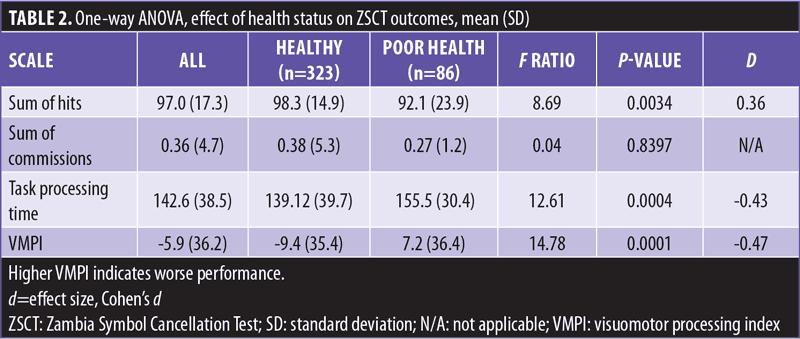
Reliability as stability after test-retest. A complete test-retest data set was obtained from a subgroup of 90 participants in apparent good health, with a mean (standard deviation [SD]) age of 143.9 (33.6) months and with 35.56 percent being male. Time interval for test-retest was 48 (±24) hours. One participant was withdrawn from the study for sudden worsening of clinical conditions preventing the execution of retest. No other cases were excluded from the final analysis for major protocol violations. Outliers were not excluded from analysis. Summary results are shown in Table 3.
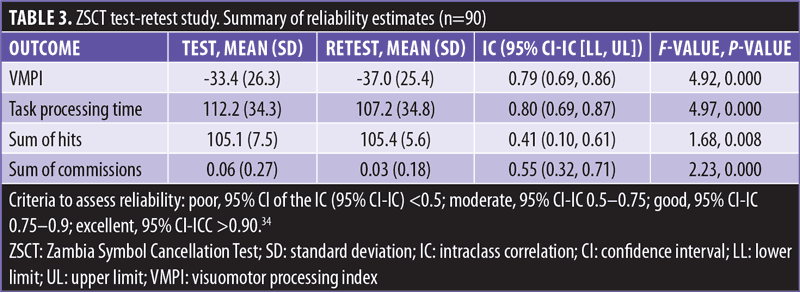
Reliability estimates were good for task processing time and poor-to-moderate for both sum of hits and sum of commissions. As such, sum of hits and sum of commissions were dropped due to unsatisfactory reliability.
VMPI as a measure of selective attention. A new outcome was derived to summarize visuo-perceptual-motor processing. A linear regression was calculated to predict sum of hits based on task processing time. We set the condition in the model that if task processing time is 0, sum of hits should be 0. Due to significant heteroscedasticity, a robust regression was applied. A significant regression equation was found [n=323, F (1, 322): 2660.82, p=0.000, R2=0.8649]. Task processing time was a significant predictor of sum of hits. Predicted sum of hits was (0.6388782*task processing time) when task processing time is measured in seconds. Predicted sum of hits increased by 0.64 for each task processing time second. The R2 value indicated that a large proportion (86.49%) of variance in sum of hits could be explained by the task processing time. We concluded that the regression model fit observed data. Predicted sum of hits was interpreted as a standard reference of effortful cognitive processing.
Then, we derived VMPI from the predicted sum of hits. VMPI was calculated for each study participant (N=409) based on the following formula: VMPI=(0.6388782*task processing time)–sum of hits. VMPI mean (SD) was -5.9 (36.2). VMPI values ranged between -107.37 (a hypothetical case in which the cancellation task was perfectively processed, with 108 hits in 1 second), and +115 (the task was poorly processed with no hits in 180 seconds). The correlation between VMPI and task processing time was as follows: n=323, r=0.93, p=0.000.
VMPI was constructed as a measure of effortful processing to deliver an accurate response exercised by the cognitive system. For example, lower VMPI scores indicated the extent to which higher accuracy of response was achieved by lower utilization of time resource.
As shown on Table 3, VMPI reliability estimate was good and acceptable for the use of the ZSCT in a clinical setting.
Effect of sex and age on VMPI. One-way ANOVA showed that VMPI mean score for the male group (n=146, mean [SD]: 6.1 [34.7]) was higher than the female group (n=177, mean [SD]: -12.1 [35.8]). The difference was not statistically significant (defined as p<0.05) between the two groups [F (1, 322): 2.31, p=0.13]. There was no effect of sex on VMPI.
Age showed a large effect on VMPI (n=323, r= -0.62, p=0.000). This was interpreted as evidence that the visuo-perceptual-motor processing effort to accurately complete the task decreased with age in a school-aged population, reflecting an increased efficiency of the cognitive system.
Effect of age and health status on VMPI. A between-groups, two-way ANOVA design was conducted to assess main and interaction effects of health status and age group on VMPI. We compared the poor health group to the healthy group. Sample characteristics are summarized in Table 1. The poor health group included children with at least one chronic health condition that was recognized as an important risk or causative factor for cognitive impairment and required a therapeutic intervention at the time of study assessment.
The diagnostic classification was based on medical review of a child’s available health information, as made available by medical history, clinical and neurological examination, instrumental diagnosis, or reporting from reliable proxies. The healthy group showed more favorable indicators of social functioning, such as the ability to speak multiple languages (i.e., more than one Zambian language, such as Bemba and Nyanja in Lusaka area, and English at school). Delayed access to school was more frequent in children with poor health status.
The effect of age was evaluated between childhood (ages 5–7 years), late childhood (ages 8–10 years), early adolescence (ages 11–13 years), and late adolescence (ages 12–17 years) groups. Cell sizes, means, and SDs for the 2 x 4 factorial design are shown in Table 4. Statistical assumptions for analysis were satisfactorily met.
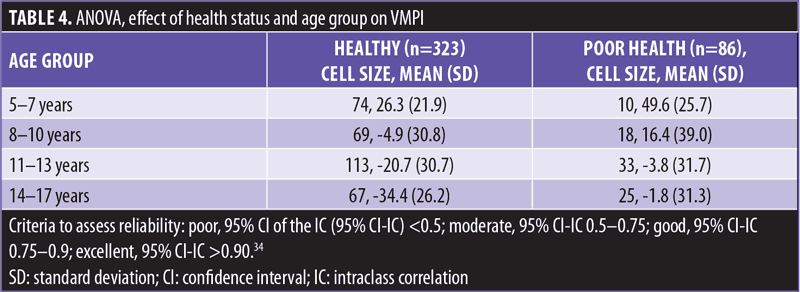
Overall, health status and age group effects were significant [F (7, 408): 33.24, p=0.0000, η2=0.367]; main effect of health status [F (1, 408): 37.79, p=0.000, η2=0.09] and age group [F (3, 408): 35.06, p=0.000, η2=0.21] was statistically significant and small, and their interaction was not significant (p=0.364).
In summary, using higher visuo-perceptual-motor processing effort to accurately complete the task was associated with poor health status. Children with a poor health condition exercised higher visuo-perceptual-motor processing effort than healthy children to achieve an accurate response.
Discussion
The ZSCT is a paper-and-pencil cognitive test with a user-friendly, simplified, and fast (test completed within 5 minutes) procedure of administration and scoring. It is intended for use in healthcare and clinical research settings to detect and monitor attention deficits over time.
The ZSCT provides a measure of selective attention, VMPI. VMPI showed satisfactory reliability when administered to a sample of Zambian school-aged children, when the purpose of the measurement was to provide an assessment of cognitive status. Stability over time was successfully demonstrated. VMPI reliability estimate at test-retest is adequate for clinical use.
VMPI measures the extent of cognitive resource utilization to achieve an accurate task response; as such, it is an indicator of the level of effort cancellation task processing requires of the cognitive system. For example, consider two children. The child who hits all 108 targets in 90 seconds has a VMPI of -50.5. As only 90 of the180 seconds allowed were used for task processing and were sufficient to satisfy the functional demand to achieve an optimal behavioral outcome (i.e., accuracy of 100% target hits), it can be concluded that a relatively low effort was exercised by the cognitive system. The second child, with comparable age, health, and sociocultural status, who hits 90 targets in 180 seconds obtains a VMPI of 25.0. Higher VMPI shows that the effort to overcome the functional demands posed by the task required a relatively higher utilization of resources (i.e., higher task processing time), only to achieve a poorer response (i.e., accuracy of 50% target hits). As VMPI reflects the modality of use of cognitive resources (e.g., efficiency) for cancellation task processing, it can be concluded that the cognitive system of the second child in this example was less efficient, as a larger effort was exercised to overcome the task processing demands.
VMPI decreases with age from childhood to adolescence, demonstrating that the efficiency of visuomotor processing and attentiveness in children increases over time. This finding is consistent with results from other studies.20,29,30 It also provides evidence supporting the validity of VMPI use in evaluating cognitive development.
VMPI is dependent on health status. Lower visuo-perceptual-motor processing efficiency is associated with poor health status. This finding is consistent with previous research suggesting health conditions as important risk factors for cognitive impairment or altered cognitive development in children.19 It also highlights the potential utility of VMPI use in evaluating the effect of health-related risks or protective factors on cognitive development.
Limitations. An important limitation of our research relates to the characteristics of the study sample, as it is not representative of the entire Zambian population, due to its composition and limited size. All the participants were from an urban area, whereas most of the pediatric population lives in rural areas. A new field trial on a larger, more representative normative sample is warranted to determine an accurate set of norm values, along with guidelines for healthcare professionals regarding the interpretation of these norm values. Future work on the ZSCT norming samples should prioritize the enrollment of a representative group of children living in rural areas, as well as a group of children who do not attend school, to minimize the risk for misdiagnosis in children with a limited exposure to educational settings.
Another limitation relates to the study design, which allowed only a partial assessment of ZSCT psychometric properties. However, results enable and provide information on next steps of the validation process. First, future research should provide supporting evidence for convergent and discriminant validity of VMPI interpretation. Notwithstanding the limited number of cognitive tests originally validated in Zambia, the use of instruments imported to Zambia and cross-culturally validated should be prioritized for these studies. Evaluations should verify the overlap of VMPI with measures of other constructs. Moderate correlations would be expected with measures of visuoconstructional ability (e.g., from copy-design tasks), episodic memory (e.g., free recall paradigms), attention (e.g., focused, divided, and sustained), and working memory (e.g., span). The relation of VMPI to measures of nonverbal intelligence, as measured by the Raven’s Progressive Matrices and the Panga Munthu Test, is of particular interest.
Second, in regard to study design, the ZSCT has potential for use in hypothesis-driven or exploratory clinical research and epidemiology. The ZSCT can help to better characterize the clinical manifestations and burden of widespread medical (e.g., malaria, human immunodeficiency virus/acquired immunodeficiency syndrome [HIV/AIDS] infection, postinfective neurocognitive complications, malnutrition, etc.) or psychosocial conditions (e.g., social deprivation) on cognitive functioning and development. The ZSCT could also be applied to clinical trial design for the purpose diagnostic classification or as evaluation outcomes (e.g., efficacy, safety, cost efficiency) of group- or population-based intervention programs. Further investigation should be undertaken to assess reliability of the ZSCT when the purpose of the measurement is prediction or monitoring of medical intervention (e.g., evaluation of a therapeutic treatment). Specifically, the responsivity to clinical change could be evaluated by a repeated measurement design.
Third, in regard to study design, study results support the validity of the ZSCT to evaluate cognitive development and the effect of different disease conditions on pediatric cognitive functioning. New research should address clinical utility and diagnostic accuracy of the ZSCT in relation to specific disease conditions.
Conclusion
The assessment of pediatric cognitive health in Sub-Saharan developing countries poses significant challenges, including the paucity of valid diagnostic tools. The ZSCT addresses the need for new psychometric tests to evaluate attention in a pediatric, school-aged population. The ZSCT is a viable and reliable instrument for cognitive testing. Study results warrant further evaluations to validate its use in healthcare and clinical research settings.
Acknowledgments
The authors wish to thank the members of the Working Group on Zambia Paediatric Cognitive Assessment Tools for their contribution to the project: Sr. Virginia Chanda, Dr. Ornella Ciccone, Mr. Kalima Kalima, Mrs. Prisca Kalyeyle, Dr. Syvia Mwanza Kabaghe, Dr. Nfewa Kawatu, Dr. Lisa Nkole, Mr. Aaron Phiri, Dr. Somwe Somwe, Ms. Mercy Sulu, and Mr. Owen Tembo.
References
- Fernald LCH, Prado E, Kroger P, Raikes A. Toolkit for Measuring Early Childhood Development in Low- and Middle-Income Countries. International Bank for Reconstruction and Development/The World Bank; 2017. https://openknowledge.worldbank.org/bitstream/handle/10986/29000/WB-SIEF-ECD-MEASUREMENT-TOOLKIT.pdf?sequence=1. Accessed 17 Jan 2023.
- Matafwali B, Serpell R. Design and validation of assessment tests for young children in Zambia. In: Serpell R, Marfo K, eds. Child Development in Africa: Views from Inside: New Directions for Child and Adolescent Development. Wiley; 2014:77–96.
- Mulenga K, Ahonen, T, Aro M. Performance of Zambian children on the NEPSY: a pilot study. Dev Neuropsychol. 2001;20(1):375-383.
- Brooks BL, Sherman EM, Strauss E. Test review: NEPSY-II: a developmental neuropsychological assessment second edition. Child Neuropsychol. 2010;16(1):80–101.
- Korkman M, Kirk U, Kemp S. NEPSY: A Developmental Neuropsychological Assessment Manual. The Psychological Corporation; 1998.
- Deregowski JB, Serpell R. Performance on a sorting task: a cross cultural experiment. Int J Psychol. 1971;6(4):273–281.
- Ezeilo B. Validating Panga Munthu Test and Porteus Maze Test (wooden form) in Zambia. Int J Psychol. 1978;13(4):333–342.
- Kathuria R, Serpell R. Standardisation of the Panga Munthu Test. A non-verbal cognitive test development in Zambia. J Negro Educ. 1998;67(3):228–241.
- Di Cesare F, Di Carlo C, Di Cesare L. Development of a Cognitive Ability Assessment Tool (CAAT) for use in paediatric clinical trials in Sub-Saharan countries. Innov Clin Neurosci. 2021;18(10–12):30–37.
- Fink G, Matafwali B, Moucheraud C, Zuilkowski SS. The Zambian Early Childhood Development Project: 2010 Assessment Final Report. Harvard University; 2012.
- Zuilkowski SS, McCoy DC, Serpell R, et al. Dimensionality and the development of cognitive assessments for children in Sub-Saharan Africa. J Cross Cult Psychol. 2016;47(3):341–354.
- James WA. The Principles of Psychology. Henry Holt; 1890.
- Lavie N, Hirst A, De Fockert JW, Viding E. Load theory of selective attention and cognitive control. J Exp Psychol Gen. 2004;133(3):339–354.
- Treisman AM. Selective attention in man. Br Med Bull. 1964;20:12–16.
- Treisman AM. Strategies and models of selective attention. Psychol Rev. 1969;76(3):282– 299.
- Posner MI. The attention system of the human brain. Annu Rev Neurosci. 1990;13:25–42.
- Goldstein EB. Sensation and Perception, 8th edition. Wadsworth; 2010:134–136.
- Stevens C, Bavelier D. The role of selective attention on academic foundations: a cognitive neuroscience perspective. Dev Cogn Neurosci. 2012;2(Suppl 1):S30–S48.
- Gall S, Müller I, Walter C, et al. Associations between selective attention and soiltransmitted helminth infections, socioeconomic status, and physical fitness in disadvantaged children in Port Elizabeth, South Africa: an observational study. PLoS Negl Trop Dis. 2017;11(5):e0005573.
- Ickx G, Bleyenheuft Y, Hatem SM. Development of visuospatial attention in typically developing children. Front Psychol. 2017;8:2064.
- Riva D, Devoti M. Discontinuation of phenobarbital in children: effects on neurocognitive behavior. Pediatr Neurol. 1996;14(1):36–40.
- Stevens C, Lauinger B, Neville H. Differences in the neural mechanisms of selective attention in children from different socioeconomic backgrounds: an event-related brain potential study. Dev Sci. 2009;12(4):634–646.
- Gall S, Adams L, Joubert N, et al. Effect of a 20-week physical activity intervention on selective attention and academic performance in children living in disadvantaged neighborhoods: a cluster randomized control trial. PLoS One. 2018;13(11):e0206908.
- Brickenkamp R, Zillmer E. The d2 Test of Attention. Hogrefe and Huber Publishers; 1998.
- Gauthier L, Dehaut, F, Joanette Y. The Bells Test: a quantitative and qualitative test for visual neglect. Int J Clin Neuropsychol. 1989;11(2):49–54.
- Katz N, Cermak S, Shamir Y. Unilateral neglect in children with hemiplegic cerebral palsy. Percept Mot Skills. 1998;86(2):539–550.
- Ickx G, Hatem SM, Riquelme I, et al. Impairments of visuospatial attention in children with unilateral spastic cerebral palsy. Neural Plast. 2018;2018:1435808. https://doi: 10.1155/2018/1435808
- Wilson B, Cockburn J, Halligan P. Behavioural Inattention Test. Thames Valley Test Company; 1987.
- Laurent-Vannier A, Chevignard M, Pradat-Diehl P, et al. Assessment of unilateral spatial neglect in children using the Teddy Bear Cancellation Test. Dev Med Child Neurol. 2006;48(2):120–125.
- Vakil E, Blachstein H, Sheinman M, Greenstein Y. Developmental changes in attention tests norms: implications for the structure of attention. Child Neuropsychol. 2009;15(1):
21–39. - StataCorp. Stata: Release 15. Statistical Software. StataCorp LL; 2017.
- Koo TK, Li MY. A guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med. 2016;15(2):155–163. Erratum in: J Chiropr Med. 2017;16(4):346.
