 by Anna Furnari, MD; Rocco Salvatore Calabrò, MD, PhD; Donatella Imbesi, MD, PhD; Francesca La Fauci Belponer, PT; David Militi, PhD; Giuseppe Gervasi, PhD; Concetta Pastura MD, PhD; and Placido Bramanti, MD
by Anna Furnari, MD; Rocco Salvatore Calabrò, MD, PhD; Donatella Imbesi, MD, PhD; Francesca La Fauci Belponer, PT; David Militi, PhD; Giuseppe Gervasi, PhD; Concetta Pastura MD, PhD; and Placido Bramanti, MD
All from IRCCS Centro Neurolesi “Bonino-Pulejo,” Messina, Italy
Innov Clin Neurosci. 2014;11(1–2):23–25
Funding: No funding was provided for the preparation of this manuscript.
Financial disclosures: The authors have no conflicts of interest relevant to the content of this article.
Key words: Electronic baropodometer instrument, Parkinson’s disease, atypical parkinsonism, gait, postural instability
Abstract
Objective: The differential diagnosis between atypical parkinsonism and Parkinson’s disease is difficult, especially in the early stage. Severe postural instability, falls, and complex gait impairments are usually confined to the later stage of Parkinson’s disease, while atypical parkinsonism patients may present a severe postural instability with consequent falls in the earlier stages.
Methods: We retrospectively studied 20 subjects with parkinsonism using clinical and baropodometric tools to give quantitative and objective data on the postural, balance, and gait disturbances.
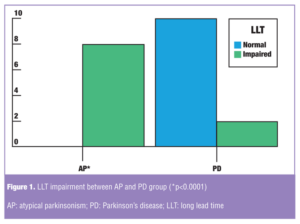
Results: The statistical analysis between atypical parkinsonism and Parkinson’s disease patients showed a significant difference in the frequency of long lead time parameter, foot area, foot load and speed, and, in particular, atypical parkinsonism patients presented a prevalent long lead time impairment (8/8 patients) when compared with Parkinson’s disease patients.
Discussion: Beside significant differences in the clinical features between the Parkinson’s disease and atypical parkinsonism, our study showed that baropodometric investigation may a valuable tool for the definition of postural and motor extrapyramidal abnormalities, permitting an earlier differentiation between atypical parkinsonism and Parkinson’s disease.
Background
Parkinson’s disease (PD) is the most common neurodegenerative cause of parkinsonism, a clinical syndrome characterized by lesions in the basal ganglia. The primary lesion of PD, which makes up approximately 80 percent of cases of parkinsonism, is degeneration of the neuromelanin-containing neurons in the brainstem, particularly those in the pars compacta of the substantia nigra. The cardinal motor signs and symptoms of PD include the pathognomonic clinical picture of resting tremor, rigidity, akinesia, and impairment of postural reflexes. Although preclinical detection of PD has been investigated, a practical, sensitive, and specific screening test is not yet available. Furthermore, in the absence of a disease-specific biologic marker, a definitive diagnosis of PD can be made only at autopsy by the presence of specific neuropathological findings including Lewy bodies. The differential diagnosis between PD and atypical parkinsonism (AP), especially in the early stage, is difficult since there are, to date, no specific markers confirming the diagnosis. Thus, it would be useful to have a simple and noninvasive tool to differentiate the parkinsonian syndromes. Severe postural instability falls and complex gait impairments are usually confined to the later stage of PD, while AP patients may present a severe postural instability with consequent falls the earlier stages.[1]
Aim of our study was to compare individuals with PD and those with AP, using clinical and baropodometric tools to give quantitative and objective data on the postural, balance, and gait disturbances of these diseases in order to facilitate the differential diagnosis.
Materials and Methods
We retrospectively studied 12 individuals with PD (median age 75, ranging 73–76 years) and eight subjects with AP (median age 70, ranging 67–72 years) followed at the IRCCS Neurolesi of Messina in Italy.
Diagnosis was performed according the Gebb’s clinical criteria and through the Unified Parkinson’s Disease Rating Scale motor subscores (UPDRS III),[2] the Hoehn and Yahr (H&Y) staging,[3] and the Mini-Mental State Examination (MMSE).[4]
The Modular Clinical Electronic Baropodometer (Diasu Company, Rome, Italy) consists of a walkway constituted by a foot-force platform placed on the floor interfaced to software for the acquisition of the load forces for each foot and of postural attitude in static and dynamic position and was used for the instrumental evaluation. The following parameters have been studied: static analysis including the plantar surface (PS) and plantar load (PL); stabilometric analysis, including the length of the ball (long lead time [LLT]/mm2); dynamic analysis, including velocity (cm/sec), semi-step length (SSL/cm), cadence (step/minutes), stance phase (SP %), swing phase (SwP %) and double support phase (DSP%). For each session at least three tests, with five-minute intervals of rest, were performed. The study was approved by the Local Ethics Committee.
Statistical analysis was performed using the SYSTAT software version 9 running on Windows, and statistical differences were detected through Chi-Square test. Statistical significance was considered at p=0.05 level.
Results
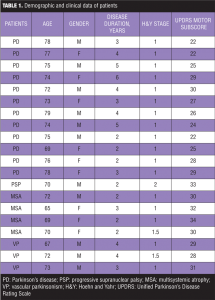
Demographic and clinical data of the two patient groups are displayed in Table 1.
PD patients had a median disease duration of 3.5 years (range 2.75–4.5), a H&Y stage range of 1 to 2, and a median UPDRS III score of 25 (range 24–28). AP patients (1 with PSP, 3 with vascular parkinsonism, and 4 with MSA-P) presented a median disease duration of 2.5 years (range 2.0–3.5), a H&Y stage range of 1 to 2, and a median score at UPDRS III of 30 (range 29–32).
All the patients were undergoing levodopa and/or dopaminergic treatment and had a MMSE greater than 24.
The statistical analysis between AP and PD patients showed a significant difference in the frequency of LLT parameter, foot area, foot load, and speed (with p<0.0001, p<0.05, p<0.01, and p<0.01, respectively). In particular, AP patients presented a prevalent LLT impairment (8/8 patients) when compared with PD patients (2/8 patients) as shown in Figure 1.
Discussion
Postural instability (PI), one of the most disabling features of the late stages of PD and of the early stages of AP, seems to be related to many factors, such as dysfunction of postural reflexes and of automatic and anticipatory adjustment, smaller peak of centre pressure, and reduced ability to integrate multisensory inputs, i.e. visual, vestibular and proprioceptive.[5]
Our study shows significant differences in the clinical features between the PD and AP group; nevertheless, baropodometric investigation (BI) was able to more accurately discriminate early parkinsonism from early PD, highlighting how patients affected by parkinsonian syndromes presented a more compromised motor framework than those with PD.
Indeed, in our study, the prevalence of impaired LLT in patients with parkinsonism could imply a very narrow cone of stability and weight distribution outside the cone usually resulting in falls. An anatomical correlate of the importance of basal ganglia (BG) in posture and gait has been demonstrated. In fact, it has been suggested that gait and balance disorders of PD patients are partially correlated with mesencephalic structures, which project efferent pathways to the lower brainstem and spinal cord and receive afferent inputs from BG, premotor and supplementary motor, cortical areas, and limbic system.6 Interestingly, AP patients often present with mediolateral instability, supporting the involvement of the superior cerebellar peduncle in gait and posture.[7,8] Abdo et al[9] showed that PD patients typically have a normal base of support during walking, and that the prevalence of patients with normal tandem gait was significantly higher in the PD group than in the AP group.[6]
In conclusion, our preliminary study suggests that BI may be a valuable tool for the definition of postural and motor abnormalities of extrapyramidal disease, permitting a more accurate monitoring and management of PD and an earlier differentiation between PD and parkinsonism. However, further and larger studies need to be performed to better characterize the earlier motor impairment in patients with AP, so to confirm our promising, though preliminary, findings.
References
1. Giladi N. Freezing of gait: clinical overview. Adv Neurol. 2001;21:191–197.
2. Richards M, Marder K, Cote L, Mayeux R. Interrater reliability of the Unified Parkinson’s Disease Rating Scale motor examination. Mov Disord. 1994;9:89–91.
3. Tsanas A, Little MA, McSharry PE, et al. Statistical analysis and mapping of the Unified Parkinson’s Disease Rating Scale to Hoehn and Yahr staging. Parkinsonism Relat Disord. 2012;18:697–699.
4. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state:” a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12:189–198.
5. Chastan N, Do MC, Bonneville F, et al. Gait and balance disorders in Parkinson’s disease: impaired active braking of the fall of centre of gravity. Mov Disord. 2009;24:188–195.
6. Grabli D, Karachi C, Welter ML, et al. Normal and pathological gait: what we learn from Parkinson’s disease. J Neurol Neurosurg Psychiatry. 2012;83:979–985
7. Ebersbach G, Sojer M, Valldeoriola F, et al. Comparative analysis of gait in Parkinson’s disease, cerebellar ataxia, and subcortical arteriosclerotic encephalopathy. Brain.1999;122:1349–1355.
8. Ondo W, Warrior D, Overby A, et al. Computerized posturography analysis of progressive supranuclear palsy. Arch Neurol. 2000:57,1464–1469.
9. Abdo WF, Borm GF, Munneke M., et al. Ten steps to identify atypical parkinsonism. J Neurol Neurosurg Psychiatry. 2006;77:1367–1369.