by Muruga A. Loganathan, MD; Manasa Enja, MD; and Steven Lippmann, MD
by Muruga A. Loganathan, MD; Manasa Enja, MD; and Steven Lippmann, MD
Dr. Loganathan is a resident in the Department of Psychiatry, Allegheny General Hospital, Pittsburgh, Pennsylvania; Dr. Enja is an observer physician in the Department of Psychiatry, University of Louisville, Louisville, Kentucky; and Dr. Lippmann is Professor, Department of Psychiatry, University of Louisville, Louisville, Kentucky.
Innov Clin Neurosci. 2015;12(5–6):38–41.
This ongoing column explores off-label or emerging treatment options, drug development trends, and theoretical concepts in the field of neuroscience.
Funding: No funding was provided for the preparation of this article.
Financial disclosures: The authors have no conflicts of interest relevant to the content of this article.
Key words. Forced normalization, epilepsy seizures, seizure disorder, seizure control convulsions, ictal disorder, ictus, psychosis, psychotic, pharmcotherapy medication
Abstract: Forced normalization is the emergence of psychoses following the establishment of seizure control in an uncontrolled epilepsy patient. Two illustrative clinical vignettes are provided about people with epilepsy that was newly controlled and followed by emergence of a psychosis; symptoms appeared only after attaining ictal control. For recognition and differential diagnosis purposes, understanding forced normalization is important in clinical practice.
Introduction
Seizure disorders and psychiatric presentations are complex, interrelated conditions. There is a higher incidence of psychosis (9%) in people with seizures, when compared to the general population (1%).[1] There is also a relationship between new ictal control and onset of psychotic symptoms; the term forced normalization refers to this clinical scenario.[2,3]
In the past, artificial induction of convulsions with camphor had been prescribed as a treatment for psychoses.[3] Similarly, electroconvulsive therapy-induced seizures can result in diminishing levels of psychosis.[4] Forced normalization is the converse of these conditions, i.e., the emergence of psychosis after the control of seizures. The precise mechanism of this phenomenon is unknown, but it can occur whether convulsions are controlled by medication, surgery, or neurostimulation.
Clinical Vignettes
Case 1. A 35-year-old man with a history of mental retardation and epilepsy was hospitalized with psychotic delusions. The patient had no previous mental illness. Since childhood, he exhibited generalized tonic-clonic, complex partial, atonic, and tonic seizures. Antiepileptic drug trials had included phenytoin, felbamate, and levetiracetam; without efficacy or side-effects, he had a corpus callosotomy 12 years ago. A recent previous video/ electroencephalogram (vEEG) recorded five generalized tonic-clonic convulsions over a five-day period, with seizures beginning simultaneously in both hemispheres.
A vagal nerve stimulator was implanted to control his epilepsy. After this, convulsions ceased once prescribed phenytoin 300mg daily, felbamate 1,200mg three times daily, and levetiracetam 1,250mg twice daily. As the seizures diminished, the patient became delusional, claiming that he was pregnant, and responding to visual hallucinations.
Once hospitalized, routine laboratory results were within normal limits. His free phenytoin level was 2.26mcg/mL, the total phenytoin was 19.1mcg/mL, and levetiracetam concentration was 13mcg/mL. Brain imaging revealed no intracranial abnormalities other than those resulting from the previous surgeries; these scanning results were similar to others reported when he was hospitalized for uncontrolled seizures earlier that year.
On this admission, the EEG illustrated a background of a well-modulated seven hertz rhythm, with awake, drowsy, and sleep recordings evidencing no epileptiform activity. Antipsychotic pharmacotherapy with quetiapine was instituted and levetiracetam was reduced to a 1,000mg twice a day. On repeat vEEG recording, the patient was confused, staring toward his right side with apparent loss of muscle tone in his upper limbs; two similar complex partial seizures were noted; thus, levetiracetam was increased back to the original dose. Subsequently, there was no further ictal activity. As an outpatient, the patient’s delusions persisted, but he remained free of convulsions.
There were many questions posed while considering the diagnosis of this patient. Is this ictal psychosis or forced normalization? The psychotic symptoms did not remit nor follow the typical time duration pattern fitting an ictal psychosis profile. Considering forced normalization, clinical seizures ceased and there were EEG changes compatible with partial seizures. Psychosis due to a general medical condition, epilepsy, was the psychiatric diagnosis.
Case 2. A 41-year-old woman was hospitalized for treatment of paranoia and recent personality change. Paranoid, confused, agitated, and wanting to jump out of a window, she required restraints for safety. Family reporting indicated that this happened whenever her epilepsy medications were adjusted; she would become overtly paranoid and argumentative. Hallucinations emerged as seizures came under control following a recent trial of lacosamide. The EEG evidenced normal background activity with no evidence of seizures. A head scan revealed a porencephalic cyst, encephalomalacia communicating with the right lateral ventricle, and a patent right-sided ventriculo-peritoneal shunt.
The past history included treated infantile meningitis and a ventriculoperitoneal shunt. She began having complex partial seizures at age seven, presenting with episodes of unusual smells and feelings of déjà vu. These progressed to loss of awareness and were associated with picking at clothing or wringing of hands. Complex partial seizures occurred several times a month. The ictal pattern changed to one of generalized tonic clonic seizures after her second pregnancy; episodes were documented on EEG recordings. Multiple medications were prescribed to treat her epilepsy. With convulsions refractory to pharmacotherapy, a vagal nerve stimulator was implanted.
Psychotic episodes followed whenever medications established control. Her family reported a cycle of psychosis, nonadherence, and recurrence of seizures for years, despite being prescribed phenytoin and valproic acid. The antipsychotic drug quetiapine increased the ictal frequency. During this admission, lacosamide was discontinued and the psychosis gradually diminished. Her epilepsy was stabilized following adjustments in the levetiracetam and antiepileptic medication regimen.
The etiology of her psychotic episodes remains unknown. It is unclear as to whether the psychoses are pharmaceutical side effects or due to establishing seizure control; the pathophysiology is undetermined. The balance between antiepileptic medications, antipsychotic drugs, ictal suppression, and psychosis requires clinical monitoring and communication between the patient, family, and physician.
Forced Normalization
The clinical vignettes evidence two individuals with difficult-to-treat epilepsy that, with ictal control and relative normalization of the EEG, developed psychiatric dysfunction during neurological stability. Such mental health issues in people with epilepsy typify forced normalization.[1] It is impossible to know whether this dysfunction was due to ictal psychosis or forced normalization. Also, the influence of anticonvulsive pharmacotherapy clouds this differential.[5]
Identifying psychoses in relation to ictal presence is a challenge. Psychoses in epilepsy have a complex psychopathology that is similar to the positive symptoms of schizophrenia and usually without negative symptoms.[6] Psychoses with seizure disorders can be further differentiated as ictal, post-ictal, and inter-ictal, depending on the timing at arousal of symptoms. When psychosis occurs during a seizure and is related to that event by EEG, that is called an ictal-psychosis. Post-ictal psychoses arise later following lucidity after a convulsion. Inter-ictal psychosis is not related in time to seizure occurrence and emerges between episodes.[7] Post-ictal psychosis most commonly appears within a week of the last seizure, whereas psychosis in forced normalization continues to manifest after normalization and with a clear state of consciousness.[8]
Diagnostic norms of forced normalization include 1) epilepsy substantiated by history and EEG recordings, 2) new behavioral disturbances, 3) reduction by half in the number of EEG spikes, 4) a one-week absence of seizures, 5) similar previous episodes, and 6) occurrence of such events on changes in antiepileptic pharmacotherapy.[1,5] This can occur with generalized and focal epilepsies. When fewer EEG spikes or normalization are not documented by EEG, this diagnosis is supported by having similar episodes in the past or recent alterations in the therapeutic regimen.
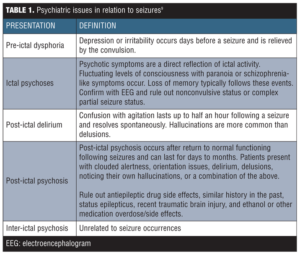
See Table 1 for psychiatric issues related to seizures.[9]
The two vignettes had differences in their presentation. The first patient’s EEG was not completely normalized, although the clinical seizures ceased. The second case evidenced EEG normalization. This deviates from the diagnostic criteria since the first person’s EEG did not stabilize, yet psychotic symptoms emerged, whereas in the second person the EEG normalized. These changes pose a challenge in establishing forced normalization diagnosis. It is important to understand what to expect clinically and to inform patients about their prognosis and close monitoring plans.
Concept
Behavioral issues in people with epilepsy have been described since the 19th century; psychosis or agitations were considered to be a part of the clinical picture.[5,10] An antagonism between schizophrenia and epilepsy was hypothesized during the development of electroconvulsive therapy.[2]
Since the 1950s, it is known that patients with poorly controlled epilepsy exhibit psychotic episodes following seizure remission, with disappearance of epileptiform activity on EEG recordings.[5] Understanding this relationship between psychosis and epilepsy remains controversial.
The term forced normalization is emergence of psychiatric symptoms on electrical stabilization of the EEG.[1] The term alternative psychosis applies to the clinical phenomenon of a reciprocal relationship between abnormal mental states and seizures that did not rely on EEG findings.[10] Paradoxical normalization describes epilepsy that is still active, but remains subcortical.
The reasons for these presentations remain unclear;[11] one explanation, however, could involve disinhibition of the limbic system after gaining seizure control. The cortex has an inhibitory function on the limbic system, but this balance becomes destabilized upon ictal control.[12] Since convulsions generally suppress psychoses, psychotic symptoms can be attenuated by medically induced seizures.[10]
Another explanation could be limbic kindling. The kindling induced by electric or neurotransmitter processes is considered to result in these behavioral changes. Repeated limbic stimulation, mainly at the amygdala, olfactory bulb, and pyriform cortex appear to create a behavioral action followed by motor responses.[1,13] The cortex, however, is more resistant than the limbic system to these stimulations. It is hypothesized that chemical kindling causes behavioral issues and electrical ones result in motor seizures.[14,15] Repeated dopamine agonist stimulation can yield psychoses and other permanent behavioral disturbances, which are also labeled as pharmacological kindling. Gaba-amino-butyric acid (GABA) potentiation at the limbic system in patients with epileptic hippocampal sclerosis can precipitate depression.[15]
Confounding Factors
The vignettes illustrate confounding factors and suggest forced normalization. Medication side effects, epilepsy surgery, and other techniques of controlling seizures, such as vagal nerve stimulation, are known to precipitate psychotic symptoms. The development of psychosis raises the question as to whether this is a post-ictal phenomena or ongoing subclinical seizures. Stabilized epileptic patients whose medication dosages are reduced and seizures reoccur are usually without psychiatric symptoms.[1] Once epilepsy is controlled, the psychiatric symptoms appear. This phenomenon is common in individuals with intellectual disability and refractory epilepsy, such as in Lenox-Gastaut syndrome. Once neurologically stabilized, they often develop psychosis and depression.[16]
Conclusion
Control of seizures does not mean absence of neuropathology, since electrical instability remains active subcortically and might supply energy for the psychotic symptoms.[3] Forced normalization can be more difficult to treat than post-ictal psychosis. Such ictal psychoses are prevented by seizure control, while inter-ictal psychosis requires a multidisciplinary approach that includes psychopharmacological management.[17] Complete control of seizures can induce psychotic symptoms and worsen quality of life. Patients and doctors must balance between good seizure control and an ability to function without overt psychopathology.[18] Clinical monitoring, EEG readings, and physician communication will aid diagnosing and treating these individuals for optimal response.
References
1. Kanner AM, Palac S. Neuropsychiatric complications of epilepsy. Curr Neurol Neurosci Rep. 2002;2(4):365–372.
2. Krishnamoorthy ES, Trimble MR. Forced normalization: clinical and therapeutic relevance. Epilepsia. 1999;40(10):57–64.
3. Bob P. Dissociation, forced normalization and dynamic multi-stability of the brain. Neuroendocrinol Lett. 2007;28(3):231–246.
4. Wilkins KM, Ostroff R, Tampi RR. Efficacy of electroconvulsive therapy in the treatment of nondepressed psychiatric illness in elderly patients: a review of the literature. J Geriatr Psychiatry Neurol. 2008;21(1):3–11.
5. Krishnamoorthy ES, Trimble MR, Sander JW, Kanner AM. Forced normalization at the interface between epilepsy and psychiatry. Epilepsy Behav. 2002;3(4):303–308.
6. Hasija D, Jadapalle SLK, Badr A. Status epilepticus and psychosis of epilepsy. Psychiatr Ann. 2012;42(1):11–13.
7. Lancman M. Psychosis and peri-ictal confusional states. Neurology. 1999;53(5):33–38.
8. Schachter SC, Holmes GL, Trenité DK. Behavioral Aspects of Epilepsy: Principles and Practice. New York: Demos Medical Publishing; 2008:489–495.
9. Trimble MR. Ictal and post-ictal psychiatric disturbances. In: Duncan JS, Sisodiya SM, Smalls JE (eds). Epilpesy 2001: From Science to Patient. Edenbridge: Meritus Communications; 2001:261–265.
10. Wolf P, Trimble MR. Biological antagonism and epileptic psychosis. Br J Psychiatry. 1985;146:272–276.
11. Wolf P. Acute behavioral symptomatology at disappearance of epileptiform EEG abnormality: paradoxical or “forced” normalization. Adv Neurol. 1991;55:127–142.
12. Pollock DC. Models for understanding the antagonism between seizures and psychosis. Prog Neuropsychopharmacol Biologic Psychiatry. 1987;11(4):483–504.
13. Adamec RE. Does kindling model anything clinically relevant? Biologic Psychiatry. 1990;27(3):249–279.
14. Post RM, Kopanda RT. Cocaine, kindling, and psychosis. Am J Psychiatry. 1976;133(6):627–634.
15. Mula M, Monaco F. Antiepileptic drugs and psychopathology of epilepsy: an update. Epileptic Disord. 2009;11(1):1–9.
16. Yagi K. Intractable epilepsy and disturbed visuomotor performance. Jap J Psychiatry Neurol. 1987;41(3):335–341.
17. Nadkarni S, Arnedo V, Devinsky O. Psychosis in epilepsy patients. Epilepsia. 2007;48(9):17–19.
18. Gobbi G, Giovannini S, Boni A, et al. Catatonic psychosis related to forced normalization in a girl with Dravet’s syndrome. Epileptic Disord. 2008;10(4):325–329.